Lithium-ion batteries are one of today's best technologies for storing electrochemical energy. They have a high energy density (energy per unit volume) and specific energy (energy per unit of weight) and a sufficiently long lifetime to allow them to be used in microelectronic devices and cars. The rise of Li-ion batteries is impressive: they have achieved astonishing commercial success in just two decades. However, further improvements are possible and this is a field in which researchers at the Paul Scherrer Institute (PSI) are working. Nevertheless, the potential of the Li-ion battery is limited chemically and it will only be possible to achieve an even higher energy density, which is of critical importance for electric mobility in particular, by using other new types of batteries.
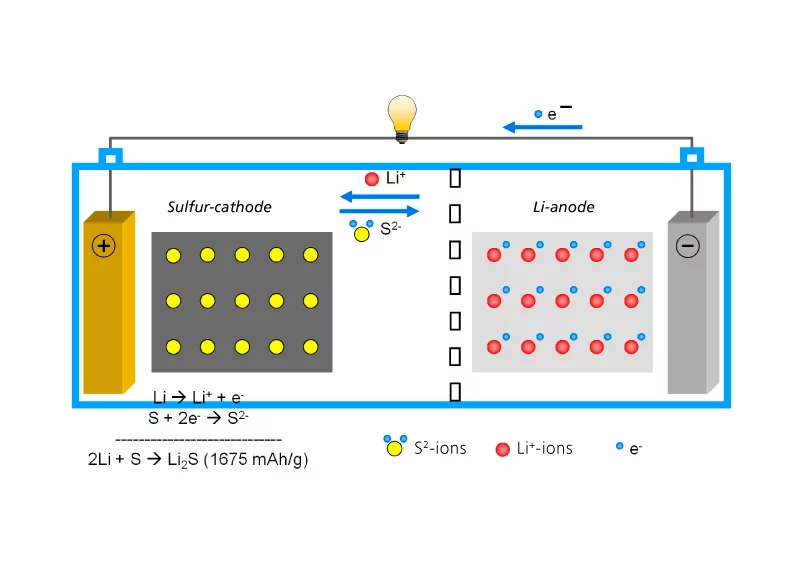
One of the most promising alternatives is the lithium-sulfur battery. In this type of battery, the anode is made of metallic lithium, while the cathode is made of a composite comprising sulfur and carbon. The lithium-sulfur battery is a “conversion” type battery, because the electrochemical reactions which take place during charging and discharging of the battery result in new chemical compounds. By contrast, lithium-ion batteries operate in accordance with the “insertion” principle. This means that lithium ions occupy spaces in the crystal structure of the cathode, without substantially changing the structure of the cathode material. Generally, the amount of energy which can be stored increases as the number of bound or inserted lithium ions increases. The higher energy density of the lithium-sulfur battery is due to the fact that each sulfur atom can bind two lithium ions, whereas, in the case of conventional battery types, less than one lithium ion is “captured” on average. The advantage of lithium-sulfur batteries is primarily their superior storage capacity (energy density), but they also excel in economic and environmental terms. Sulfur is a relatively inexpensive and abundant raw material, unlike expensive elements, such as cobalt, which are used in lithium-ion batteries. This is important because material costs make up a very large proportion of the total cost of a battery. From an environmental perspective, lithium-sulfur batteries also make it possible to avoid the use of heavy metals. According to Petr Novák, Head of the Electrochemical Energy Storage Section of the Electrochemistry Laboratory at PSI, no toxic chemicals are used in the manufacture of lithium-sulfur batteries.
The biggest challenges for the lithium-sulfur battery
The path to market for the lithium-sulfur battery is still lined with some challenges. A fundamental difficulty is the fact that sulfur is an electrically insulating material. Therefore, it has to be embedded in a matrix of electrically conductive carbon in the cathode. Otherwise, the lithium ions would only reach the sulfur atoms on the surface of the cathode. Adding carbon creates an electrically conductive structure, which allows access to sulfur atoms over the entire body of the cathode. However, the necessary addition of the carbon simultaneously makes the battery heavier and bulkier, which can lower the energy density. Another problem, which is specific to the lithium-sulfur battery, is the “polysulfide shuttle”. This is the name given by experts to the interaction of the cathode and the lithium anode via chemical reactions with polysulfides – intermediates from the chain of electro-chemical reactions which power the discharging and charging of the battery. These polysulfides are soluble in the liquid organic electrolytes of the battery cell and can therefore migrate from the cathode, where they originate, to the anode. They then react with lithium atoms on the surface of the anode. This results in the surface becoming “passivated”, as researchers call it, because the lithium atoms, which are now incorporated in the passivation layer, are no longer available to store energy. This causes the cell to “age”; that is to say, it loses much of its storage capacity after only a few charge and discharge cycles. One objective of many studies carried out in the development of the lithium-sulfur battery is therefore to prevent the polysulfides detaching themselves from the cathode, moving to the anode and accumulating there. While previous attempts to gain control of this process have resulted in slight improvements, these usually mean a higher complexity of the battery structure and, consequently, higher production costs. PSI researchers, led by Petr Novák and working with the German chemical company BASF, are looking for solutions which can be implemented industrially, with the focus on profitability. They have reported some initial success.
Better performance through the use of polymers
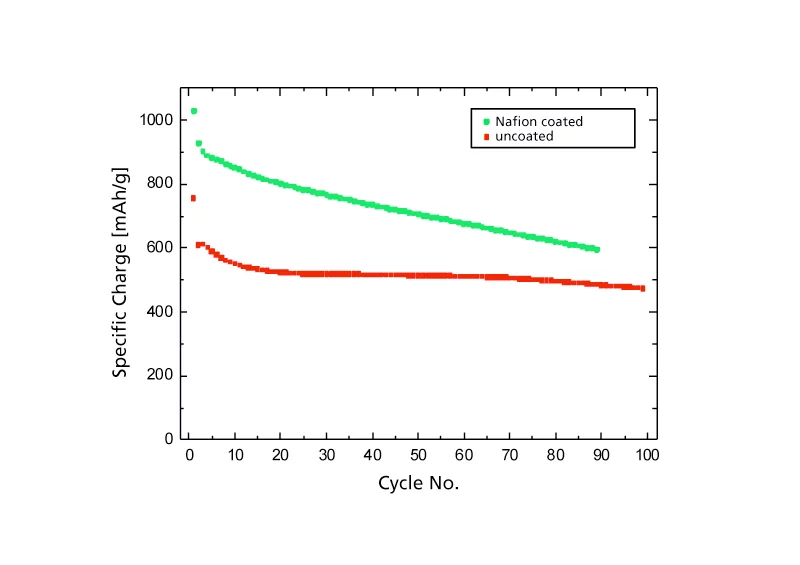
In one study, the scientists investigated the influence of different binder materials on the performance of the electrodes. A binder material is a substance which acts as a “glue” in order to hold the components of an electrode together. Binder materials are necessary, because the charging and discharging of a battery usually leads to volume changes in the electrodes. The researchers discovered that when Teflon is used as a binder material, very high values can be achieved for the energy density of a lithium-sulfur battery. Almost 70 per cent of the total storage capacity is maintained, even after 100 charge and discharge cycles. “Teflon has proved to be an excellent binder material for the cathodes of lithium-sulfur batteries. This shows that low-cost materials and relatively simple battery assemblies can lead to very good results. Our results are comparable with those of other published concepts which rely on expensive materials or complex battery architectures,” comments Novák. The researchers have not yet identified the reason for the positive effects of Teflon. Apparently, Teflon reinforces the contacts between the sulfur and the electrically conductive material in which it is embedded. The PSI researchers also suggest a connection with the water-repellent properties of Teflon which, as is well known, is used in frying pans. Their conjecture is that the “hydrophobicity” of Teflon impedes electrolyte’s access to the electroactive material sulfur, therefore preventing the polysulfides to dissolve in the electrolyte. This would result in slowing down the ageing of the battery. The researchers discovered further improvements to the battery performance when the electrodes were coated with the polymer Nafion. However, there is still no conclusive scientific explanation for this either. Nevertheless, the PSI scientists' work shows that the promising lithium-sulfur battery is an industrially viable option – rather than a “pie in the sky”, as previously thought.
N.B.: The researchers of Petr Novák's section are not only exploring the lithium-sulfur battery. Their expertise covers all of today's industrially established lithium battery types, and extends to other innovative concepts in the research and development stage.
Additional information
Section Electrochemical Energy StorageOriginal Publication
Influence of different electrode compositions and binder materials on the performance of lithium–sulfur batteriesHolger Schneider, Arnd Garsuch, Alexander Panchenko, Oliver Gronwald, Nicole Janssen, Petr Novák, Journal of Power Sources, DOI: 10.1016/j.jpowsour.2011.12.061