The PSI Laboratory for Synchrotron Radiation and Femtochemistry develops and applies X-ray based tools to study chemical processes with high spatial and temporal resolution.
Lab News & Scientific Highlights
Imaging electrical switching of ultraefficient memory devices
SLS illuminates how electrical switching in layered materials works
Swiss X-ray laser reveals the hidden dance of electrons
New X-ray technique at SwissFEL shows how electrons act together – with the potential to show why quantum information slips so easily away.
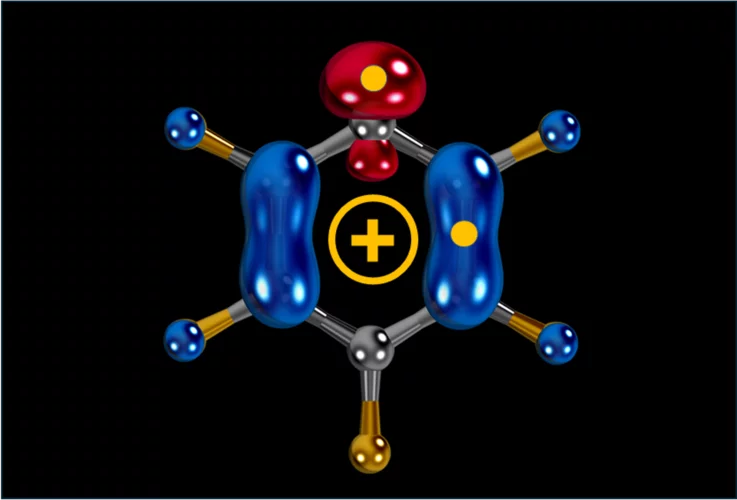
Carbocation, diradical, and superelectrophile in one molecule?
The pentafluorophenyl cation (C₆F₅⁺) breaks these rules with a borderline “crazy” reactivity.