Schweizerisch-Finnisches Forschungsteam klärt Struktur einer wichtigen Ansatzstelle für Krebsmedikamente
Um wachsen zu können, muss ein Krebstumor von Blut- und Lymphgefässen durchzogen sein, die ihn mit Sauerstoff und Nährstoffen versorgen und durch die der in der Zelle entstehende Abfall entsorgt werden kann. Um sich diese Versorgung zu sichern, senden Tumore Botenstoffe aus, die umliegende Gefässe veranlassen, sich zu verzweigen und in den Tumor hineinzuwachsen. Einen wichtigen Schritt zum besseren Verständnis der molekularen Vorgänge bei der Gefässbildung in Tumoren haben nun Forschende des Paul Scherrer Instituts und des Biomedicum in Helsinki, Finnland gemacht. Sie haben die Struktur der Bindungsstelle aufgeklärt, an der sich ein Botenstoffmolekül mit dem entsprechenden Rezeptor auf der Zelloberfläche verbindet. Durch diese Verbindung wird das Wachstum der Lymphgefässe angeregt. Dieses Wissen kann in der Zukunft für die weitere Entwicklung neuer Medikamente verwendet werden, die diese Rezeptoren gezielt blockieren. So können diese Mittel das Wachstum der Gefässe verhindern und den Tumor aushungern. Die Ergebnisse erscheinen in der Woche vom 18. Januar in der Online-Ausgabe der Zeitschrift der Amerikanischen Akademie der Wissenschaften (PNAS – Proceedings of the National Academy of Sciences of the United States of America).
Vor ca. 20 Jahren hat der amerikanische Mediziner Judah Folkman eine neuartige Tumortherapie vorgeschlagen, bei der durch gezielte Blockierung des Wachstums der Tumorblutgefässe der Tumor ausgehungert und so indirekt an seiner Ausbreitung gehindert würde. Ein auf diesem Konzept bestehender Therapieansatz wurde in der Zwischenzeit entwickelt und wird in der Klinik angewendet. Um dieses Verfahren optimieren zu können, ist es wichtig, die genauen molekularen Vorgänge zu verstehen, die hinter der Bildung der Gefässe stehen. Dazu haben die Forschenden aus der Schweiz und aus Finnland nun einen entscheidenden Beitrag geleistet.
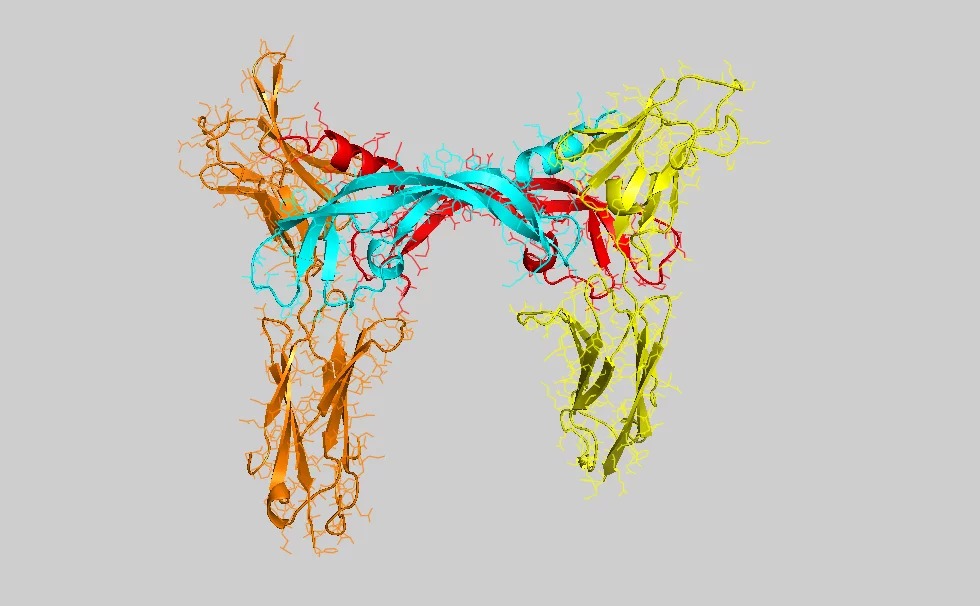
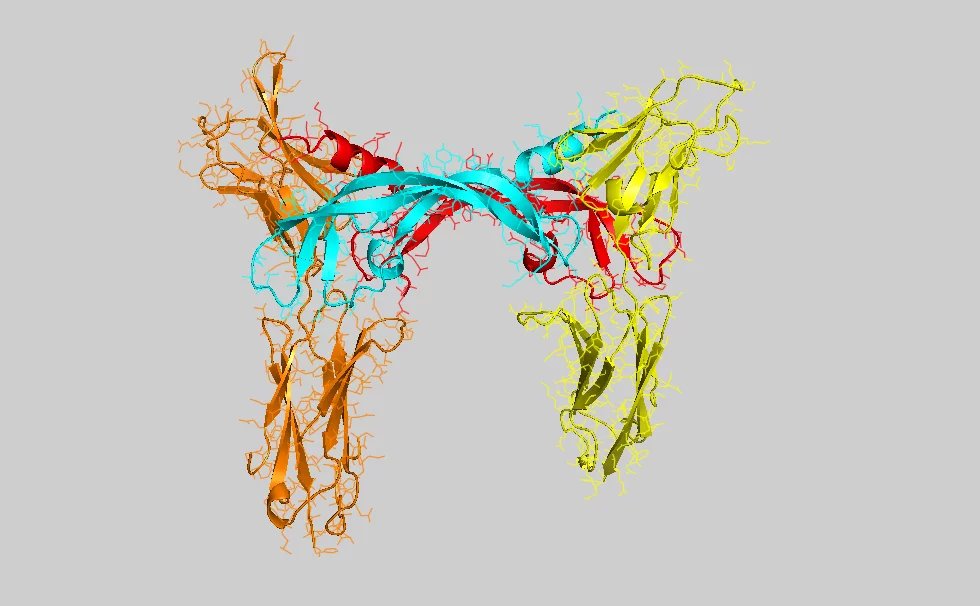
Moleküle, die für das Wachstum von Gefässen verantwortlich sind, werden von Fachleuten mit der Abkürzung VEGF (vascular endothelial growth factor) bezeichnet. Das untersuchte Molekül, das das Wachstum von Lymphgefässen anregt, heisst VEGF-C. Ein VEGF-Molekül ist biochemisch gesehen ein Protein, also ein hochkomplexes Biomolekül bestehend aus tausenden von Atomen. Es wird aktiv, in dem es sich mit dem Ende eines anderen Proteins (dem Rezeptor), verbindet, das in der Membran einer lebenden Zelle eingebaut ist. Die Bindung von VEGF an den Rezeptor bewirkt eine Veränderung der Struktur dieses Membranproteins und die daraus folgende Strukturänderung wirkt sich innert Sekunden auch auf der Innenseite der Zellmembran aus und löst dort chemische Reaktionen aus, die zur Veränderung der Zellen führen. Im hier untersuchten Fall wird die Zelle angeregt, sich zu teilen und so am Wachstum neuer Blut- und Lymphgefässe teilzuhaben.
Damit die Kombination von Signalmolekül und Rezeptor richtig funktioniert, müssen beide die richtige einmalige Molekülstruktur haben – das heisst tausende von Atomen müssen korrekt im dreidimensionalen Raum angeordnet sein. Um diese Struktur im Detail zu bestimmen, haben die Forschenden die Moleküle an der Synchrotron Lichtquelle Schweiz SLS des Paul Scherrer Instituts untersucht. Dieser Teilchenbeschleuniger mit 288 Metern Umfang erzeugt besonders intensives Röntgenlicht, mit dem unter anderem die Struktur komplexer Proteine aufgeklärt werden kann. Dazu wird das Verfahren der Proteinkristallographie genutzt, bei dem viele solcher Moleküle in einer regelmässigen Form angeordnet (kristallisiert) und anschliessend mit dem Röntgenlicht durchstrahlt werden. Dabei wird ein Teil des Lichtes in bestimmte Richtungen abgelenkt. Aus diesen Ablenkrichtungen können die Forschenden dann die detaillierte Struktur des Proteins bestimmen.
Das am PSI laufende Projekt, das zur Publikation in PNAS führte, wird vom PSI, vom Schweizerischen Nationalfonds SNF und der Schweizerischen Krebsliga unterstützt.
Über das PSI
Das Paul Scherrer Institut entwickelt, baut und betreibt grosse und komplexe Forschungsanlagen und stellt sie der nationalen und internationalen Forschungsgemeinde zur Verfügung. Eigene Forschungsschwerpunkte sind Festkörperforschung und Materialwissenschaften, Elementarteilchenphysik, Biologie und Medizin, Energie- und Umweltforschung. Mit 1300 Mitarbeitenden und einem Jahresbudget von rund 260 Mio. CHF ist es das grösste Forschungsinstitut der Schweiz.
Kontakt / Ansprechpartner:
Prof. Dr. Kurt Ballmer-Hofer, Labor für Biomolekulare Forschung, Paul Scherrer Institut, 5232 Villigen PSI, SchweizE-Mail: kurt.ballmer@psi.ch, Telefon: +41 56 310 4165 [Deutsch, Englisch]
Dr. Andrea Prota, Labor für Biomolekulare Forschung, Paul Scherrer Institut, 5232 Villigen PSI, Schweiz
E-mail: andrea.prota@psi.ch, Telefon: +41 56 310 5160 [Deutsch, Englisch, Italienisch, Französisch]
Originalveröffentlichung:
Structural determinants of growth factor binding and specificity by VEGF receptor 2Veli-Matti Leppänen, Andrea E. Prota, Michael Jeltsch, Andrey Anisimov, Nisse Kalkkinen, Tomas Strandin, Hilkka Lankinen, Adrian Goldman, Kurt Ballmer-Hofer, and Kari Alitalo
PNAS Early Edition, January 18, 2010