Each cell in the human body contains a network of extremely fine filaments: the cytoskeleton. Contrary to what the name suggests, the cytoskeleton is far more than just a support structure, and performs many vital functions. This makes it a promising area for research in biology and medicine, providing numerous starting points for novel therapies.
Every living thing consists of at least one cell, the basic unit of all life. Most plants and animals are made up of a multitude of cells – around 30 trillion in the case of humans, for example. Even the single cell is an enormously complex structure. An analogy is sometimes used to make it easier to understand how the cell works: the conceptual model of a city state. The cell nucleus, which contains most of the genetic material – that is, the blueprints for the cell’s essential building blocks – forms the government district, where the rules are set that determine how the whole entity functions. Ribosomes are like factories that build everything needed in the city, according to the building plans stored in the control centre – the genes – including tens of thousands of types of different proteins. These proteins, as well as nutrients, signalling molecules and other substances delivered from outside the cell, are transported in so-called vesicles, small droplets, from one place to another – like parcels handled by the postal service. The transporters are so-called motor proteins. They carry the parcels to their destinations – the various facilities across the city.
Besides the government district of the cell nucleus, there are also the mitochondria. Like power plants, they produce the fuel adenosine triphosphate (ATP) from the incoming nutrients, thus providing the energy that keeps everything running. There is a mail order company that prepares goods from the factories for transport – called the Golgi apparatus. Control stations on the city wall (cell membrane) and the gates (membrane channels and transporters) perform entry control for external goods. The police (peroximones) arrest and take away unwanted intruders, often in cooperation with the rubbish collectors (lysosomes) that dispose of waste material.
The basic prerequisites for the functioning of the cell are the previously mentioned goods transport and information exchange. Both take place mostly on the streets of the city, which connect everything in an expansive network. Parts of the so-called cytoskeleton correspond to this road network in the cell. Among other things, this is responsible for the transport of cell components along defined routes and consists of three main components: microtubules, actin filaments and intermediary filaments. Microtubules are like main roads where most of the exchange of biomolecules and other “goods” takes place. They also play an essential role in the proliferation of cells through cell division. For this reason, researchers at PSI have specialised in unveiling the secrets of these microtubules.
Investigating molecular processes
“Such dynamic structures and processes are highly complex and therefore can only be clarified with great difficulty at high resolution,” says Michel Steinmetz, head of the Laboratory for Biomolecular Research at PSI. “So far we understand only a fraction of all the molecular processes in our body, since the crucial biology happens on a tiny scale that has so far largely eluded our technical possibilities for comprehensive investigations.”
But this could change in the near future. Modern high-resolution imaging methods such as cryoelectron microscopy and X-ray crystallography made possible by the large research facilities such as those at PSI, put researchers in a position to precisely observe cell structures and their dynamic interactions with other biomolecules and substances – in atomic detail and exact to the nanosecond. This not only deepens our understanding of the organism and the molecular processes that take place within it, but also opens the door to the development of new medicines and treatments for patients with abnormal cell functions. Diseases such as cancer, Covid-19, malaria and Alzheimer’s could become easier to treat.
Vital dynamics
Microtubules consist mainly of two types of tubulins – proteins that in turn are made up of around 450 chained amino acids: alpha and beta tubulins. The two proteins form pairs and link together into thin tubes. Each of these is no more than 25 nanometres wide; a normal human hair is around 2,000 times thicker.
The network of microtubules is very dynamic, as if the streets were constantly being shortened, extended or rebuilt. Guided by various binding partners and enzymatic mechanisms, the tubulins grow and die just as plants do over the years. In timelapse mode, however: the microtubules grow from a so-called microtubule organisation centre – their root, essentially, which is located in the cell nucleus – and proceed in a certain direction by constantly docking with new tubulins. But whole rows of them also continuously fall back again, only to regrow in a different direction if possible. Often the microtubule dissolves completely after just a few minutes. If it hits a target structure and becomes a component of a solid new road, it stabilises itself and lasts longer. “This dynamism of the cytoskeleton is uniquely important for the most diverse processes in the body,” Michel Steinmetz says. For example in mitosis and meiosis – cell division, which is critical for the organism’s growth and reproduction. The cytoskeleton forms the so-called spindle apparatus and pulls apart the chromosomes, the carriers of the genetic material. In this way the DNA from the mother cell is evenly distributed between the two daughter cells. In addition, the cytoskeleton performs important functions in wound healing, the differentiation of stem cells and the networking of neurons in the brain.
Important approaches for medicine
On this dynamic road network, various motor proteins act as transporters. Dyneins, for example, take on the transport of vesicles, as well as cell organelles and other particles from the outer areas, to the centre of the cell and thus can also introduce pathogens. They take up their cargo piggyback by molecular coupling and carry it along the microtubule road as if on legs – at a rate of more than 100 steps per second. Given the length of their stride, which amounts to roughly three micrometres. In some nerve cells, whose axons can be more than a metre long, transport can take up to 12 days.
Now if viruses or parasites manage to circumvent the cell membrane’s intake controls, they too take this taxi in the direction of the cell nucleus. If just one of these invaders goes unnoticed by the cell’s own immune system, made up of peroxisomes and lysosomes, it can smuggle in its own genetic material. Then the cell produces viruses in addition to the body’s own proteins, and the infection takes hold.
Understanding this system down to the smallest detail in terms of structural biology – the dynamics of the microtubules, the movement of the motor proteins, the coupling and uncoupling processes of the body’s own proteins with foreign substances – has now become possible with modern imaging methods. “With facilities such as the Swiss Light Source SLS and the X-ray free-electron laser SwissFEL, PSI has a strong presence in this field,” Steinmetz says. And it has already scored several successes.
Finding puzzle pieces that fit
Researchers at the Laboratory for Biomolecular Research have more precisely described 27 docking sites for active substances on tubulin, 11 of which were completely unknown before. These can be thought of as indentations in the amino acid chains – which themselves are tangled together like a ball of yarn – into which some molecular compounds fit more or less perfectly, like pieces of a jigsaw puzzle. These sites are also known as binding pockets. The researchers carried out a crystallographic fragment screening. They immersed 800 tubulin crystals individually into a solution consisting of 800 different molecular fragments that are commercially available as standard active ingredients.In this way they were able to identify 56 fragments that nestled in the binding pockets, at least temporarily. “So these would be potential building blocks for a new active agent because they bind to the tubulin,” explains Andrea Prota, a scientist in the Laboratory for Biomolecular Research.
For example, there are substances that disrupt the dynamics of tubulin and thus arrest cell division. The commonly used cancer drug Taxol® curbs the proliferation of tumours and can even make them shrink. However, Taxol® is originally a natural compound derived from the bark and needles of the Pacific yew tree. Since this tree species grows in a limited geographic range and because its Taxol®- content is low, the substance needs to be produced synthetically for widespread use. This in turn is very difficult because of the enormous size of the natural molecule. On top of that, Taxol® provokes undesirable side-effects. “Structural biology now allows us to replace such substances with smaller, tailor- made molecules that only have the necessary properties to bind to the tubulin,” Prota says. “They also have a new mechanism of action, are easier to handle and with a bit of luck hopefully exhibit fewer side-effects.”
Once a fitting fragment has been found, its residence time on the tubulin needs to be increased. The longer an active agent stays bound – sooner or later even molecules that fit will be released again – the more effective it can be. The better a substance fills the binding pocket, the longer it should remain in it. So now the researchers search for other fragments to supplement the first one in order to produce a combination that fills the binding pocket optimally. To do this, they feed the measured characteristics of the discovered binding pocket and the first fragment into the computer and calculate which combination of fragments offers the optimal fit. It’s like a jigsaw puzzle for which the computer knows all the pieces in detail and can suggest the right ones.
This is how the experts at PSI and IIT, the Instituto Italiano di Technologia in Genoa, developed the active agent Todalam. They measured one of the discovered binding pockets with atomic precision using SLS and then, in the computer, combined three fitting fragments into a single molecule that perfectly filled the pocket – as subsequent measurements confirmed. In cell cultures, the researchers then proved that Todalam, like Taxol©, actually does disrupt the dynamics of microtubules in cells and thus stops their proliferation. “Our studies showed for the first time that it is possible to develop tailor-made small molecules, assembled from fragments, which inhibit microtubules,” Steinmetz says. “In addition, Todalam is easy to recreate chemically and to produce in large quantities. Now it is important to show if it can be used to develop a novel cancer drug.”
In a new study published at the start of this year, a research group led by Jörg Standfuss produced a film, using the SwissFEL, created virtually in ultra- high temporal and spatial resolution. The film shows how a drug – in this case the cancer drug combretastatin A-4, currently being tested in clinical trials – detaches from the binding pocket and how both the active ingredient molecule and the pocket deform in the process. “A deeper understanding of these processes, which we have now managed to visualise for the first time, improves our possibilities of customising new active ingredients in a way that increases the attachment time and efficacy,” explains Standfuss, who works at PSI’s Laboratory of Biomolecular Research.
A targeted effect is required
One difficulty remains, however: how to design new active agents in such a way that they only target and block diseased cells, not healthy cells – since this is largely what provokes undesirable side-effects.
Research is already farther advanced in the effort to defend against single-celled parasites trying to infect the body. Here too, researchers at the Laboratory for Biomolecular Research are applying the principle of using tubulin dynamics to stop the hostile cells and prevent them from dividing.
Compared with fighting proliferating cancer cells, the advantage is that the tubulins of the parasites are constructed somewhat differently from those in humans. This could already be seen in their amino acid sequences and was confirmed through high-resolution imaging. PSI researchers Natacha Gaillard and Ashwani Sharma have analysed tubulins from the cells of the inconspicuous ciliated animal Tetrahymena thermophila. They are more or less identical to the tubulins of the malaria pathogen of the genus Plasmodium and the toxoplasmosis pathogen Toxoplasma gondii. On the atomic level, the researchers identified clear structural differences between these tubulins and those of humans. Then, by screening a database of active agents, they identified five candidates for substances that could bind to this form of tubulin in a targeted manner. In tests, one proved to be a hit. Gaillard and Sharma dubbed this compound Parabulin.
Cooperation partners at the University of California in the USA tested Parabulin in cultures of human cells that were infected with Toxoplasma gondii. And in fact, the parasite could no longer multiply, while the human cells were as good as unaffected. “The Parabulin was able to prevent the invasion of the cell by toxoplasmodia as well as their proliferation within the cell,” Gaillard reports. This would probably work with other parasites, and that is currently being tested. “For that, though, it seems clear that different Parabulin variants must be customised through structural biology.”
To continue down this path, Gaillard and Sharma have applied for a patent and founded the start-up ASTRA Therapeutics to develop species-specific Parabulins for targeted treatment of malaria, toxoplasmosis and other diseases caused by parasites.
Meanwhile at PSI, Gaillard and Sharma are working to advance fundamental research on this topic: “The more precisely we know the processes through which parasites misuse the cytoskeleton and its functions for their purposes, the more accurately we can target treatments,” Gaillard says. For example, they want to find out which protein a parasite uses to dock, as well as where and how, on the microtubule. Then, binding could perhaps be blocked on the side of the parasite or the host, or maybe they could be prevented from finding each other.
Propellers and conveyor belts made of microtubules
Researchers associated with Takashi Ishikawa, a group leader in PSI‘s Laboratory for Nanoscale Biology, are on the trail of other severe diseases and new therapies. They are studying cilia and flagella. These are thread-like appendages found on many cells, which likewise are made up of microtubules and thus, in a way, represent extensions of the cytoskeleton; they too fulfil many kinds of functions in the body.
We know them, for example, from the sperm, which is propelled by a flagellum like a fishtail when it is looking for a way to the egg. Some protozoa, such as the green alga Chlamydomonas, have two flagella that they use for locomotion. The mucous membranes in the human throat and nose are covered with carpets of such threads. In such cases they are called cilia, “but they are not really different from flagella in detail,” Ishikawa says. They move rhythmically back and forth and transport inhaled pollutants bound in the mucus, like a conveyor belt, away from the lungs and towards the trachea and throat. The harmful substances are swallowed and disposed of in the stomach. Inside the nose, olfactory cells also transport scent molecules in this way. Brain cells transport brain fluids in the same way. Cilia even play an important role in the development of embryos: here their movement serves as a kind of gyroscope that is crucial for the differentiation of the organs and their asymmetric distribution in the body. For example, the gyroscopic motion can sometimes run in the opposite direction. In patients affected by this, the organs are arranged in reverse; for example, the heart beats on the right. This is known as situs inversus.
There are countless diseases associated with defective functioning of cilia. For example, it can render both men and women infertile: men, because the sperm don’t move forwards properly; women, because the eggs in the fallopian tube are likewise transported by cilia insufficiently. Brain damage and various lung diseases are also among the socalled ciliopathies. Primary cilia dyskinesia (PCD), which afflicts around half a million people worldwide, not only affects the airways but in half of the cases is also accompanied by situs inversus.
It all comes down to protein defects. More than 400 genes are involved in the movement of cilia – that many have been discovered so far. “But exactly how the proteins control this is still unknown,” Ishikawa says. He would like to change that.
Mysterious bending
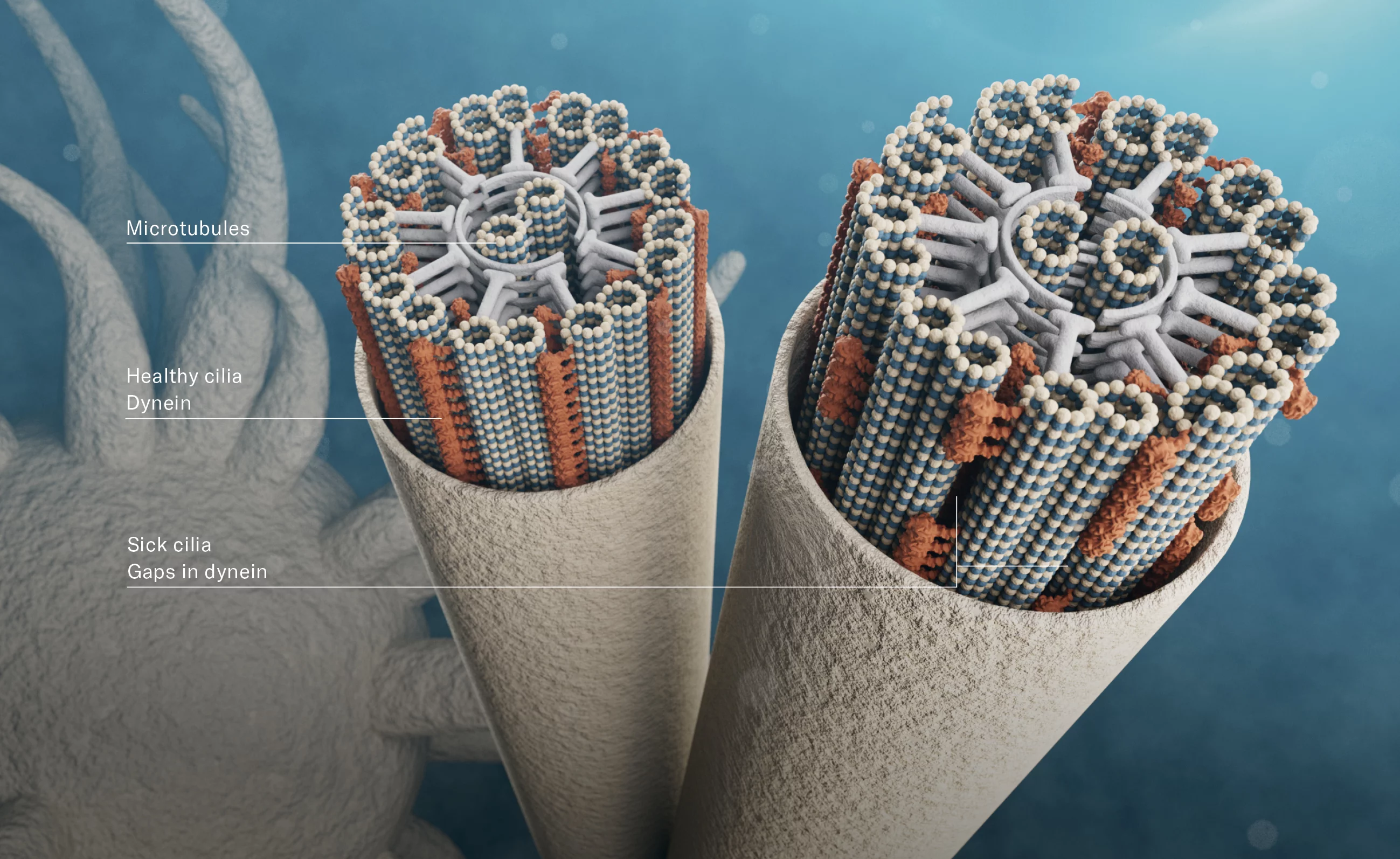
Cilia consist of a tubular framework of microtubules: two individual microtubules in the centre are surrounded by nine doublets, each made up of two microtubules fused together. Radial proteins hold it all together – just as the spokes of a bicycle wheel connect the hub with the rim and tyre. Between the outer microtubules, along the entire length, are dynein proteins – those motor proteins that travel on the microtubules. Investigations at PSI have now shown that with ATP, the fuel adenosine triphosphate, as their energy source, they cause the individual doublets to push back and forth lengthwise – similar to hands rubbing against each other. “However, we have yet to discover exactly how this gliding movement causes the cilia to bend,” Ishikawa says.
In a new study using cryo-electron microscopy, his team showed that after the dynein protein complex has cleaved the ATP, it looks different than it did before. There is a molecular rearrangement of its building blocks, the amino acids. “We suspect this is the driving force behind it,” Ishikawa says. But his team has only observed one small part of the entire ciliary apparatus. “We still need to examine many other parts to identify connections and to characterise the overall mechanism.” Presumably, the structural changes in the dynein differ along the cilia, so that they influence each other in a certain mechanism and cause the movement.
It will take years before the picture is complete. But the studies already offer a concrete benefit today: the PSI researchers are cooperating with the Inselspital of the University of Bern, which is providing tissue samples from PCD patients. Ultrahigh-resolution images of their cilia show that there are often large gaps in the dynein covering of the filaments, and in some the dynein is even completely absent. At present, it can be several years between the first symptoms of PCD and the point when a patient experiences acute respiratory problems, goes to the doctor and receives the diagnosis. This means valuable time is lost for potential early intervention. “This could be shortened significantly through routine visual inspection of the cilia,” Ishikawa says.
New means for targeted corrections
These examples show that research into the molecular processes in our body using the new imaging methods promises great progress. At the same time, it is humbling. “When we see how delicately linked, dynamic and finely balanced all these processes are, we researchers are in awe,” says Michael Steinmetz. “What biology is now revealing to us, piece by piece, is difficult for us to comprehend in all its complexity – a true wonder. All the same, the new methods available to us here at PSI enable us to understand a bit more about it – and possibly to make more targeted corrections in the event that something goes wrong in the system.”
Author: Jan Berndorff
Copyright
PSI provides image and/or video material free of charge for media coverage of the content of the above text. Use of this material for other purposes is not permitted. This also includes the transfer of the image and video material into databases as well as sale by third parties.