Experimente am SwissFEL sollen helfen, genau zu verstehen, wie sich Stoffe in einer Reaktion ineinander umwandeln. Ein Schwerpunkt werden katalytische Reaktionen sein, die zahllose Anwendungen haben. Die Forschung wird Wege zu energiesparenden Industrieprozessen und umweltfreundlichen Energieträgern aufzeigen.
In der Natur wandeln sich Stoffe ständig ineinander um – sie lösen sich auf, neue entstehen. In der chemischen Industrie werden gezielt zahllose Substanzen hergestellt, die als Treibstoffe, Kunststoffe, Medikamente oder Düngemittel wesentliche Bestandteile unseres Alltags geworden sind. Auch wenn diese Vorgänge zum Teil schon seit Jahrzehnten erfolgreich genutzt werden, sind sie oftmals nicht im Detail verstanden. Für die Praxis bedeutet das, dass diese Reaktionen nicht mit maximaler Effizienz angewandt werden können – der Energieverbrauch ist unnötig hoch, es wird nur ein Teil der Ausgangssubstanzen umgewandelt oder es entsteht übermässiger Abfall.
Zu klein und zu schnell für bisherige Verfahren
Die Vorgänge während einer chemischen Reaktion sind unvorstellbar schnell: auch wenn es Tausendstelsekunden braucht, bis aus den Ausgangsstoffen die Endsubstanzen entstanden sind, dauern die einzelnen Bewegungen der Moleküle und Atome oft nur einige Femtosekunden. Und auch wenn trickreiche Experimente zum Teil jetzt schon erlauben herauszubekommen, in welchen Kombinationen die Atome während der Reaktion kurz zusammenfinden – die Wege von der einen Zwischenstufe zur nächsten blieben bisher verborgen. Das wird sich dank des SwissFEL nun ändern: Seine Blitze dauern so kurz, dass sich die Molekülbestandteile nur wenig bewegen, während sie mit einem solchen Blitz beleuchtet werden. Und weil die Blitze aus Röntgenlicht bestehen, kann man auch die Lage der Atome sichtbar machen. Damit kann man die an der Reaktion beteiligten Teilchen mitten in der Bewegung ablichten – ähnlich wie man einen Sportler mit kurzer Belichtungszeit mitten in der Bewegung fotografiert.
Vertraute Vorgänge verbessern – neue Vorgänge entwickeln
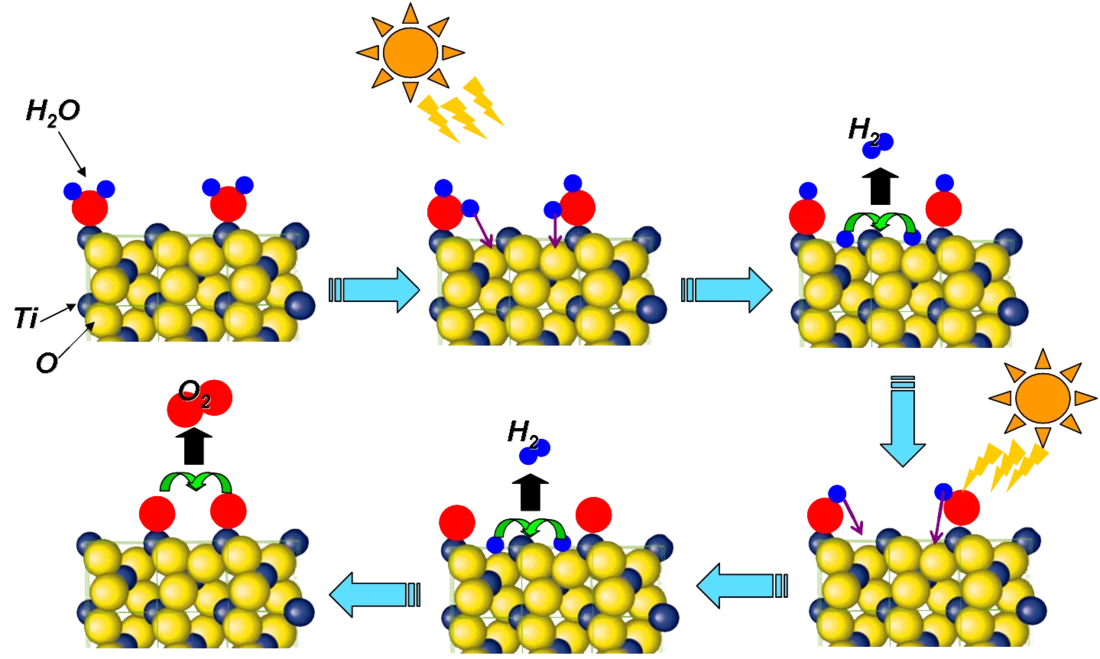
Einen Schwerpunkt der Untersuchungen zu chemischen Vorgängen am SwissFEL werden katalytische Reaktionen darstellen. Diese spielen eine grosse Rolle in der chemischen Industrie – rund 80 % aller chemischen Produkte, werden mithilfe solcher Reaktionen hergestellt. Es lohnt sich also, diese Reaktionen genauer zu verstehen und effizienter zu gestalten, weil damit grosse Energieeinsparungen möglich werden. Ein klassisches Beispiel für eine solche katalytische Reaktion ist die Ammoniaksynthese, die die Grundlage für die Herstellung von künstlichem Stickstoffdünger bildet.
Die Forschung am SwissFEL soll es möglich machen, neuartige Prozesse zu entwickeln, die auf katalytischen Reaktionen beruhen. Ein Stichwort dabei ist „künstliche Fotosynthese“. In diesem Verfahren sollen mithilfe von Sonnenenergie aus Kohlendioxid und Wasser Methan oder Methanol hergestellt werden, die als Grundlage für chemische Produkte und Treibstoffe genutzt werden könnten. Mit diesem Verfahren ist es möglich, Kohlendioxid, das bei der Verbrennung entsteht, gleich wieder für die Produktion neuen Brennstoffs zu verwenden. Nutzt man so hergestellte Brennstoffe, erhöht sich die Menge an CO2 in der Atmosphäre nicht. Zurzeit sind die entsprechenden Prozesse für eine industrielle Anwendung noch zu ineffizient – Experimente am SwissFEL sollen Wege zu effizienteren Abläufen zeigen.
Stoffumwandlung mit Unterstützung – katalytische Reaktionen
Das Besondere an einer katalytischen Reaktion ist, dass neben den Substanzen, die umgewandelt werden, noch eine weitere beteiligt ist – der Katalysator. Dieser wird zwar nicht verbraucht, ist aber unerlässlich, wenn die Reaktion effizient ablaufen soll. So kann es sein, dass man sehr viel Energie braucht, um die Bindungen zwischen den Atomen der beteiligten Moleküle aufzubrechen. Verbinden sich die Moleküle aber mit einem Katalysator – oftmals an der Oberfläche metallischer Teilchen von der Grösse weniger Nanometer– wird die Bindung innerhalb des Moleküls deutlich schwächer und lässt sich leichter auftrennen. Wie gut das funktioniert, hängt stark vom detaillierten Aufbau des einzelnen Katalysators ab. Die Arbeiten am SwissFEL werden helfen zu verstehen, wie ein Katalysator beschaffen sein muss, damit die einzelnen Reaktionen effizient ablaufen.