Der SwissFEL wird sehr kurze und sehr intensive Blitze aus Röntgenlicht mit Lasereigenschaften erzeugen und damit neue Einblicke in das Innere von unterschiedlichsten Materialien ermöglichen. Jede einzelne Eigenschaft dieses speziellen „SwissFEL-Lichts“ trägt zu den experimentellen Möglichkeiten der Anlage bei. Der SwissFEL soll 2016 in Betrieb gehen.
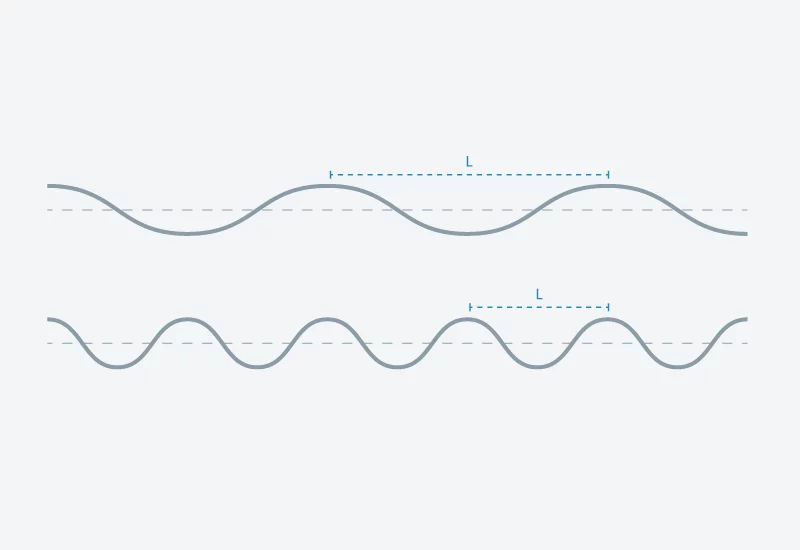
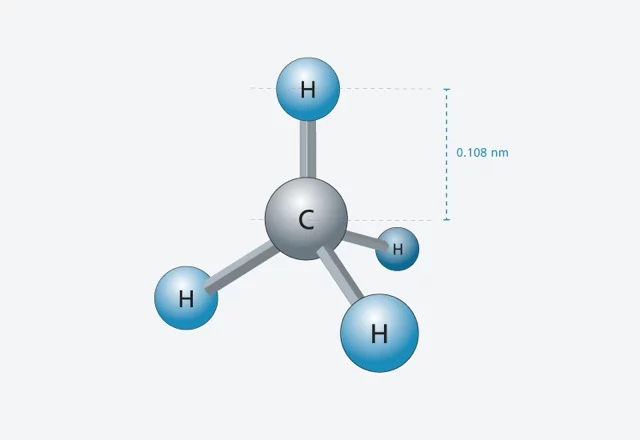
Im Prinzip ist Röntgenlicht von ähnlicher Natur wie sichtbares Licht oder Radiowellen – genau wie diese ist es eine Form elektromagnetischer Strahlung. Und wie diese ist Röntgenlicht eine Welle (siehe Infokasten). Die verschiedenen Strahlungsarten unterscheiden sich aber in der Wellenlänge – ein für die Anwendung entscheidender Unterschied. Für ein Experiment am SwissFEL fertigt man von einem Untersuchungsgegenstand eine Probe des Materials an, in dessen Inneres man schauen möchte. Je nach Art der Probe wird sie mit einem bezüglich Wellenlänge entsprechenden Licht durchleuchtet. Man kann messen, wie sich das Licht auf dem Weg durch den Untersuchungsgegenstand verändert hat und daraus auf die Struktur der Probe schliessen. Dabei werden Details sichtbar, die ungefähr so gross sind wie die Wellenlänge der Welle. Und harte Röntgenstrahlung wie sie der SwissFEL liefern wird, hat eine Wellenlänge, die etwa so gross ist wie der Abstand zwischen zwei Atomen in einem Molekül. Somit wird es möglich werden, die Strukturen von Molekülen im Detail zu entschlüsseln.
Wieso die Wellenlänge so wichtig ist, kann man leicht anhand von Wasserwellen veranschaulichen. Eine Messung mithilfe von Wellen kann man sich so vorstellen, dass man gewissermassen das Untersuchungsobjekt den Wellen in den Weg stellt und beobachtet, wie das Hindernis die Wellen verändert. Aus dieser Veränderung schliesst man auf die Form des Hindernisses. Im Meer verändern grosse Objekte wie Schiffe, Hafenmolen oder grössere Felsen sichtbar das Muster der Wasserwellen. Kleine Steine oder Sandkörner, die sehr viel kleiner sind als der Abstand zwischen zwei aufeinanderfolgenden Wellenbergen hinterlassen hingegen kaum eine Spur.