Eine neuartige Polymermembran aus dem Paul Scherrer Institut PSI hat im Labortest eine längere Haltbarkeit als die besten kommerziell erhältlichen Pendants gezeigt. Der Durchbruch gelang dank Modifizierung eines preisgünstigen Kunststofffilmes durch Bestrahlung und anschliessendes „Aufpfropfen“ funktioneller Komponenten. Der so veränderte Kunststoff hält nicht nur lange – er könnte die Herstellungskosten der Membran um 50 bis 80 Prozent senken. Anwendung finden könnte die Membran etwa in Wasserstoffbrennstoffzellen oder in Elektrolyseuren zur Wasserstoffherstellung aus Wasser.
Wasserstoffbrennstoffzellen, wie sie am PSI erforscht und entwickelt werden, könnten das Ziel einer motorisierten Mobilität ohne Nebenwirkungen für die Umwelt zur Wirklichkeit machen. In Autos eingesetzt, würden solche Zellen Strom für den Antrieb und nur Wasserdampf als Abgas erzeugen. Doch ein paar technische und ökonomische Hürden stehen diesem Traum noch im Wege. Ein Eckpunkt betrifft die Haltbarkeit der unter harschen Bedingungen funktionierenden Zelle, insbesondere der darin als selektive Trennwand verwendeten Elektrolytmembran. Andererseits müssen die Herstellungskosten noch deutlich sinken, wenn sich die Brennstoffzelle im hart umkämpften Automobilmarkt durchsetzen soll. Beiden Zielen sind Forscher des PSI nun nähergekommen. Sie haben eine Kunststoffmembran entwickelt, die den Vergleich mit ihren besten Pendants am Markt nicht scheuen muss.
Teflon und Polyethylen in einem
Ausgangsmaterial für die PSI-Membran ist der Kunststoff ETFE (Ethylen-Tetrafluor-Ethylen), ein Material, das wie jedes Polymer aus einer langen Kette von Kohlenstoffatomen als Grundgerüst besteht. An diesem Grundgerüst hängen chemische Verbindungen, die dem Polymer seine besonderen Eigenschaften verleihen. Bei ETFE wechseln sich entlang der Kohlenstoffkette jene Verbindungen ab, die jeweils die Kunststoffe Polyethylen und Teflon ausmachen. ETFE vereint deshalb die Vorzüge dieser beiden Kunststoffe, ohne ihre Nachteile zu erben: es ist leicht zu verarbeiten wie Polyethylen sowie chemisch und thermisch stabil wie Teflon.
Für elektrochemische Anwendungen muss das ETFE dennoch modifiziert werden. Insbesondere muss es ionenleitfähig gemacht und seine Gasdurchlässigkeit verringert werden. Beide Eigenschaften muss nämlich jedes Material mitbringen, das beispielsweise als Elektrolytmembran in einer Wasserstoffbrennstoffzelle zum Einsatz kommt. In einer solchen Brennstoffzelle wandern die Wasserstoffionen (Protonen) von einer Elektrode zur anderen und reagieren dann mit Sauerstoff, um Wasser und elektrischen Strom zu erzeugen. Gase, die in der Zelle vorkommen, dürfen aber nicht miteinander in Berührung kommen. Sonst könnte es zu unkontrollierten Reaktionen kommen. Deshalb braucht es eine Membran, die Protonen durchlässt, Gase aber zurückhält.
Vorsprung durch Strahlung
Modifiziert wird ETFE durch ein Verfahren, das Forscher „Strahlungspfropfen“ nennen. So wie im Gartenbau eine wertvolle, aber schwache Pflanze auf den Grundstock einer robusteren aufgepfropft wird, nutzt man hier das Gerüst von ETFE als Basis, auf die chemische Verbindungen aufgepfropft werden, die ETFE besondere Funktionalitäten verleihen. So wird quer zu den ETFE-Ketten ein Styrolderivat eingewoben, das die Ketten miteinander vernetzt und so das Gerüst mechanisch stabiler macht. Sulfonsäure kommt ebenfalls dazu und gibt ETFE die gewünschte Ionenleitfähigkeit. Mit Methacrylnitril schliesslich erhält ETFE eine wirksame Gasbarriere. Vor dem Aufpfropfen muss man das ETFE-Gerüst aber erst für die veredelnden Anhängsel empfänglich machen. Dazu bestrahlt man das Polymer mit Elektronen. Die Strahlung spaltet einzelne Atome von der Polymerkette ab und an diesen Bruchstellen können Radikale, also besonders reaktionsfreudige Moleküle entstehen. Weil die Bestrahlung in Luft geschieht, können dabei auch Peroxide entstehen, die ebenfalls bereitwillig und rasch reagieren. Diese aktiven Zentren sowie die Radikale dienen dann als Schnittstellen für das eigentliche "Aufpfropfen".
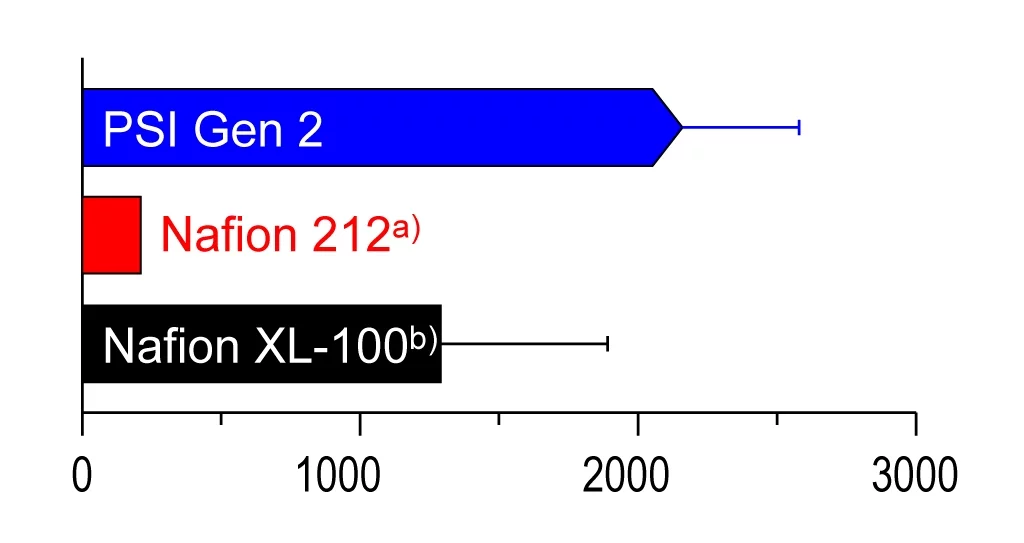
Länger haltbar als marktübliche Membranen
In Haltbarkeitstests im Labor hat sich die so funktionalisierte ETFE-Membran bereits bewährt. Zwei von drei Membranen blieben nach 2400 Stunden Testbetrieb im für Fahrzeuge typischen Belastungszyklus intakt. Damit überbietet die PSI-Membran die besten am Markt vorhandenen Membranen aus dem Kunststoff Nafion in Sachen Langlebigkeit.
Neben der überlegenen Stabilität machen potenziell erhebliche Kosteneinsparungen die PSI-Membran sehr attraktiv. Weil ETFE in der Anschaffung deutlich günstiger ist als das für solche Membranen marktübliche Nafion, rechnet man bei optimierter industrieller Herstellung mit einer Kostensenkung um 50 bis 80 Prozent. Längerfristig, wenn die Stückzahlen entsprechend steigen, würden die Herstellungskosten gemäss einer Studie der amerikanischen Firma General Motors sogar um 90 Prozent fallen.
Text: Leonid Leiva
Kontakt / Ansprechpartner
Dr. Lorenz Gubler, Leiter der Gruppe Membranen und Electrochemische Zellen,Labor für Elektrochemie, Paul Scherrer Institut,
Telefon: +41 56 310 26 73, E-mail: lorenz.gubler@psi.ch