Show filters
PILATUS4 detector arrives at PXIII
On April 4, 2025, a Dectris Pilatus4 2M detector was successfully installed at beamline PXIII. In the coming weeks, this new detector will be used to measure the first macromolecular crystallography (MX) experiments using the SLS 2.0 machine.
Thank You SLS
Our beamline scientists look back on 22 years of brilliant science made possible by the Swiss Light Source SLS.
Progress of the X06DA-PXIII beamline upgrade: First light in the optics hutch
On June 7, 2023, the PXIII project team successfully shone the first light into the optics hutch at the upgraded X06DA-PXIII beamline. It is an essential first step for testing new hardware and software solutions that will be implemented at SLS2.0.
SDU: Software for high throughput automated data collection at SLS MX
The Smart Digital User (SDU) software for unattended data collection has been deployed at the macromolecular crystallography beamlines at the Swiss Light Source.
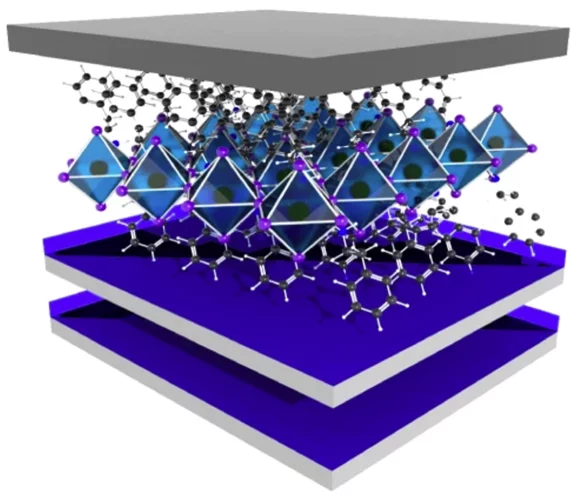
Lighting up the appealing world of hybrid perovskites
Researchers from Italy, in collaboration with the Paul Scherrer Institut, successfully used the macromolecular crystallography beamline X06DA-PXIII at the Swiss Light Source to characterize promising perovkites materials used in solar cells and other photodetector devices.
Protein distancing
PSI researchers have developed a new method to attach proteins to the surface of virus-like particles.
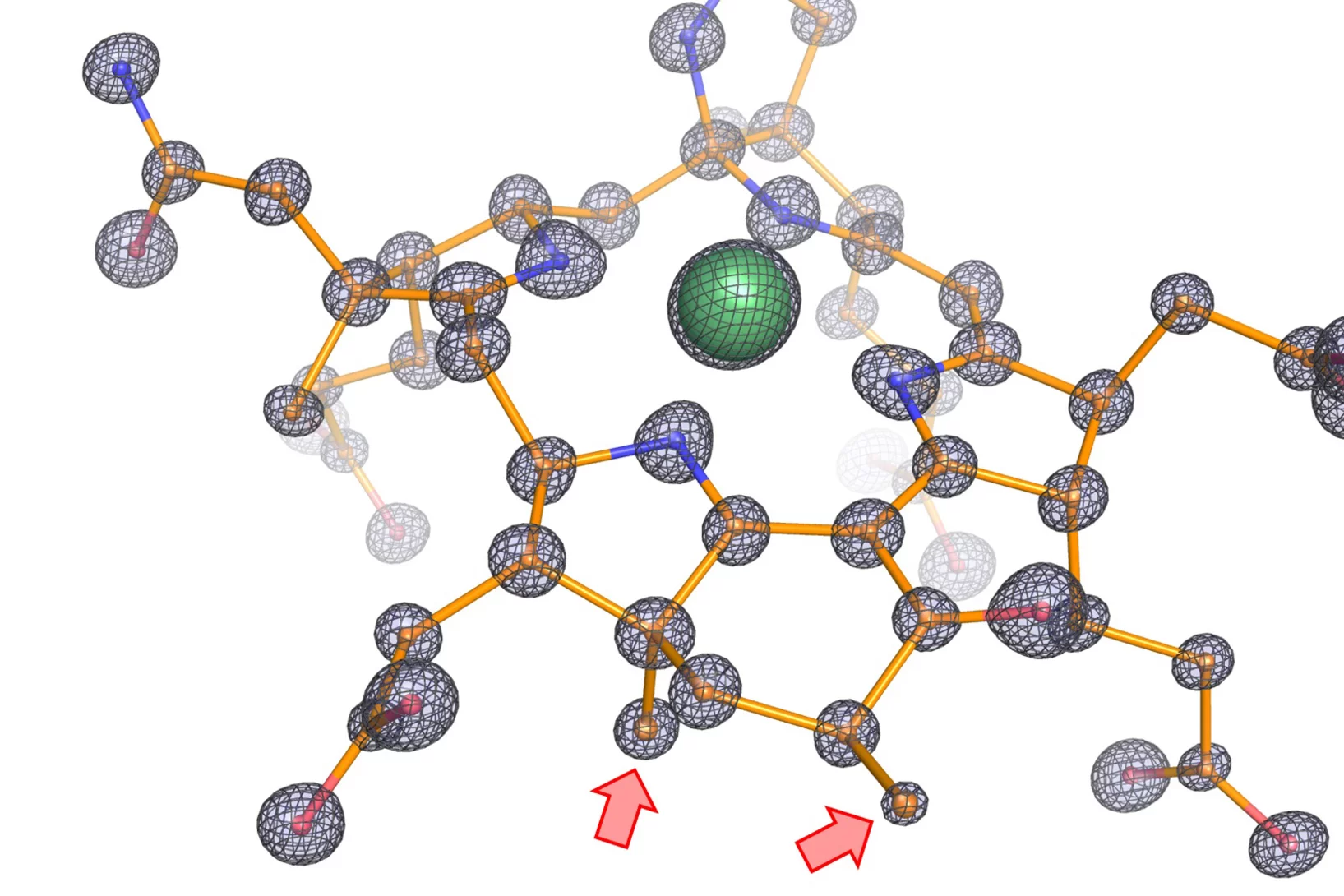
How ethane-consuming archaea pick up their favorite dish
Scientists decode the structure of the enzyme responsible for the ethane fixation by – beside others – using the SLS.
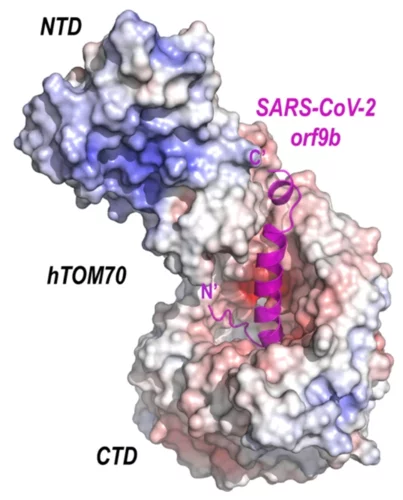
Crystal structure of SARS-CoV-2 Orf9b in complex with human TOM70 suggests unusual virus-host interactions
In a study published in Nature Communications, researchers at the NHC Key Laboratory of Systems Biology of Pathogens in Beijing, China, in collaboration with the Paul Scherrer Institut characterize the interactions of SARS-CoV-2 orf9b and human TOM70 biochemically, and they determine the 2.2 Å crystal structure of the TOM70 cytosolic domain with a bound SARS-CoV-2 orf9b peptide.
How remdesivir works against the coronavirus
Researchers at Goethe University Frankfurt, in cooperation with the PSI have probably discovered another, previously unknown mechanism of action of the antiviral remdesivir.
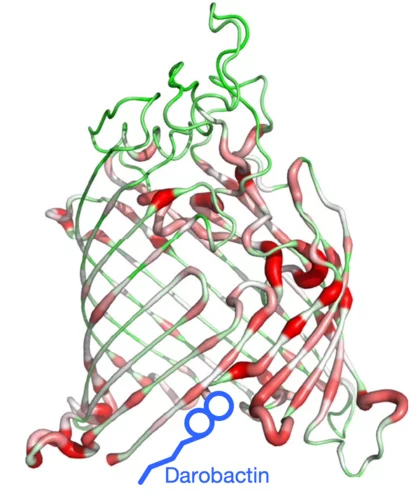
Combating antimicrobial resistance
In a study addressing the global health threat of drug resistance, researchers at the Biozentrum, University of Basel, have revealed how a new antibiotic, Darobactin, binds to the external membrane of gram-negative bacteria.
First MX results of the priority COVID-19 call
The Dikic group at the Goethe University in Frankfurt am Main, Germany has published the first results following the opening of the "PRIORITY COVID-19 Call” at SLS.
SLS MX beamtime update
Update of the SLS MX beamline operation during the COVID-19 period