MX in a nutshell
Macromolecular crystallography (MX) is a technique used to determine the three-dimensional structure of biological macromolecules, such as proteins, DNA, or RNA, through X-ray diffraction methods.
The MX Facilities at PSI
The Macromolecular Crystallography group is responsible for three beamlines at the Swiss Light Source, the Cristallina-MX endstation at SwissFEL, and the Crystallisation Facility.
Scientific Highlights
Sharper, smaller, brighter: Improved Focusing at PX III after the SLS 2.0 Upgrade
The first experimental results from the upgraded X06DA-PX III beamline after the SLS 2.0 upgrade show measurable improvements of the focusing capabilities. Comparing the focus size before the SLS upgrade and afterwards, the benefit of the new storage ring becomes immediately evident.
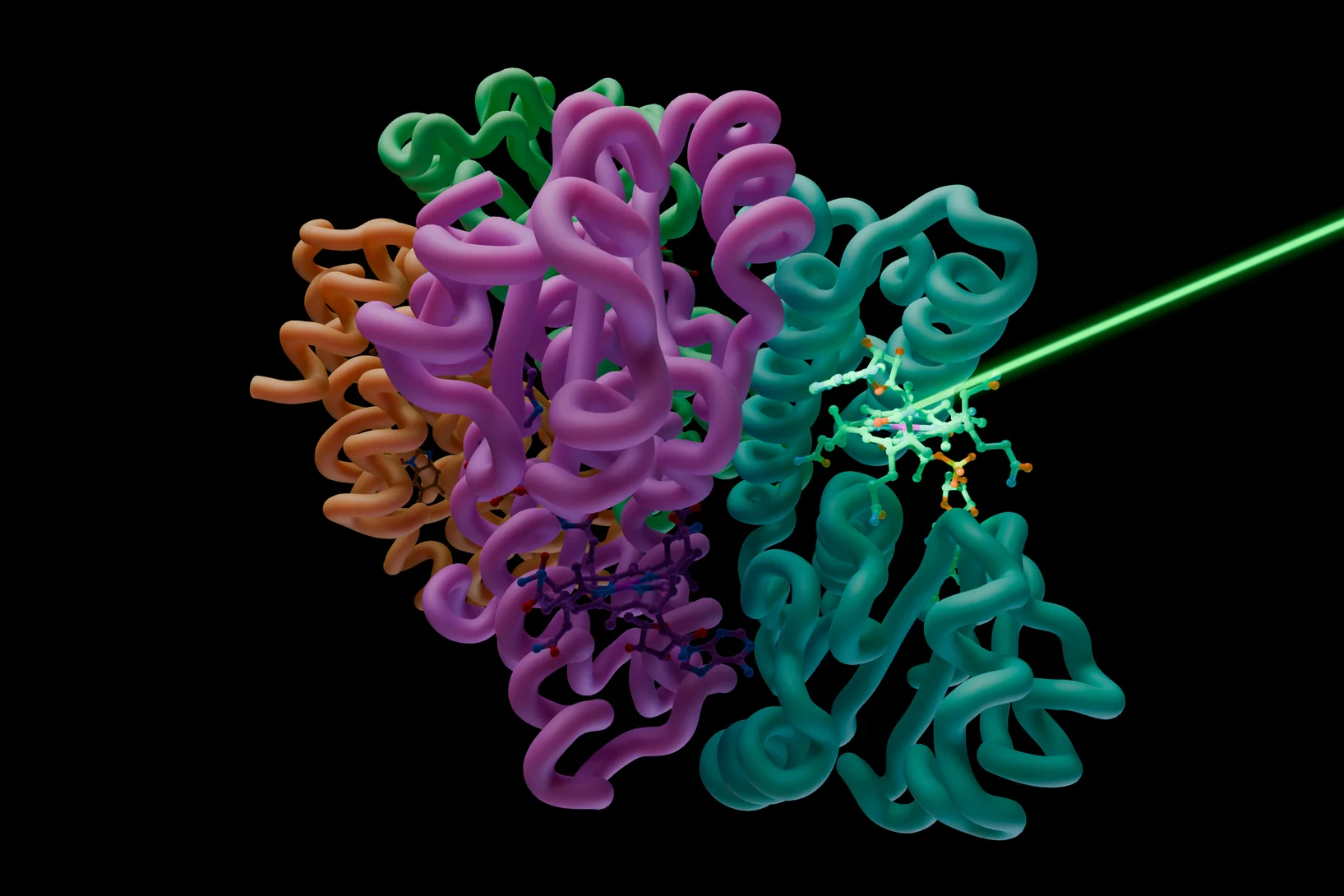
Filming a vitamin B12 photoreceptor in action
SwissFEL shows the molecular events that occur when a vitamin B12 photoreceptor absorbs light
X-raying auditory ossicles – a new technique reveals structures in record time
Using a bone, PSI researchers have demonstrated how the structures of biological materials can be determined on scales from nanometres to millimetres in a very short time.