Manipulating microscopic objects with sound—without waste or contamination.
Acoustic tweezers use sound waves to move and deform tiny objects, including living cells, without touching them. We’ve redesigned the platform so it’s both cleaner and more affordable: a reusable surface-acoustic-wave (SAW) chip mates to a disposable microfluidic cartridge, cutting per-experiment costs and minimizing cross-contamination.
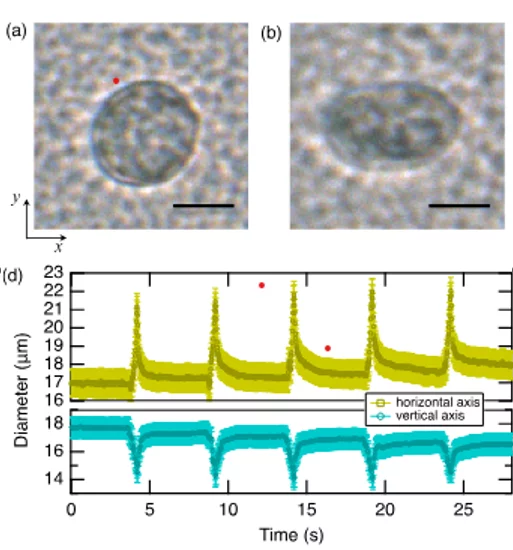
By tuning the drive to coupled chip–channel resonances near 50 MHz, we generated standing waves delivering pressures up to ~2.1 MPa. Those fields captured and gently compressed fast-swimming Tetrahymena and human cells, enabling controlled, repeatable manipulation. Time-resolved imaging let us quantify how cells deform, opening label-free routes to probe cell mechanics.
Strong, well-controlled acoustic fields in a clean, swappable cartridge make it practical to study soft materials and biological sampleswith less hassle—and to compare results across days, users, and labs.
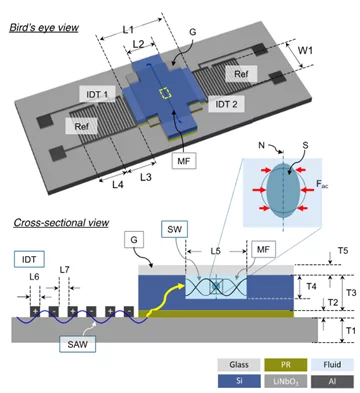
We realized such acoustic tweezers in a hybrid SAW–silicon design that concentrates acoustic energy into the channel. For chip assembly, we have developed a standardized coupling and alignment scheme. As a result, we were able to keep pressure delivery consistent. The device performance was tested by tracer-based calibration to establish the drive settings to in-channel acoustic pressure.
We successfully applied the developed hybrid acoustic tweezers to induce transient viscoelastic deformation of biological cells and to trap motile cells.
The work was recently published in IEEE Transactions on Ultrasonics, Ferroelectrics, and Frequency Control and it was featured on the front cover of the August issue of the journal.
The research was primarily conducted as a doctoral thesis of Dr. Shichao Jia at the Swiss Nanoscience Institute (SNI) PhD School and was supervised by Dr. Soichiro Tsujino (PSI) and Argovia-Professor Roderick Lim of Biozentrum at the University of Basel.
Contact
Original Publication
-
Jia S, Guo H, Lim RYH, Tsujino S
Surface-acoustic-wave-driven acoustic tweezing in a silicon microfluidic chip
IEEE Transactions on Ultrasonics, Ferroelectrics, and Frequency Control. 2025; 72(8): 1005-1014. https://doi.org/10.1109/TUFFC.2025.3581642
DORA PSI