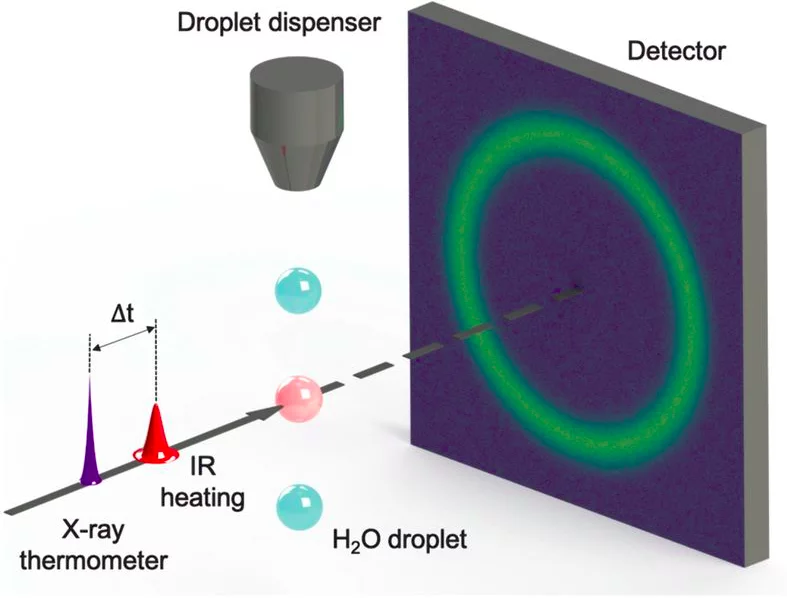
Knowledge of the temperature dependence of the isobaric specific heat (Cp) upon deep supercooling can give insights regarding the anomalous properties of water. If a maximum in Cp exists at a specific temperature, as in the isothermal compressibility, it would further validate the liquid–liquid critical point model that can explain the anomalous increase in thermodynamic response functions. The challenge is that the relevant temperature range falls in the region where ice crystallization becomes rapid, which has previously excluded experiments.
The pump/probe setup at SwissFEL Bernina was used for ultrafast calorimetry measurements, faster than the crystallisation to ice. A temperature jump was induced in micrometer-sized water droplets by an infrared laser pulse, after supercooling the water by evaporation into vaccuum down to -45°C. At this temperature droplets can remain non-frozen for short time and fingerprints of their liquid structure can be measured in X-ray scattering patterns thanks to ultrashort and bright SwissFEL X-ray pulses. Sufficient delay between the infrared laser pulse and X-ray pulses allowed measurements at constant pressure in order to determine the Cp. A sharp increase in Cp, from 88 J/mol/K at 244 K towards a maximum of about 218 J/mol/K at 229 K was observed. The Cp maximum is at a similar temperature as the previously measured maxima of the isothermal compressibility and correlation length and therefore support the liquid–liquid critical point model. The excess entropy and self-diffusion coefficient of water was estimated from the Cp measurements and show that these properties decrease rapidly below 235 K.
The experiment has been the first during the user operation phase at Bernina which started in 2019. The results of this study have been recently published.
Contact
Dr. Henrik Till Lemke
Group Leader Bernina
Paul Scherrer Institute, Forschungsstrasse 111, 5232 Villigen PSI, Switzerland
Telephone: +41 56 310 4982
Email henrik.lemke@psi.ch
Original Publication
Enhancement and maximum in the isobaric specific-heat capacity measurements of deeply supercooled water using ultrafast calorimetry
Pathak H, Späh A, Esmaeildoost N, Sellberg JA, Kim KH, Perakis F, Amann-Winkel K, Ladd-Parada M, Koliyadu J, Lane TJ, Yang C, Lemke HT, Oggenfuss AR, Johnson PJM, Deng Y, Zerdane S, Mankowsky R, Beaud P & Nilsson A
Proceedings of the National Academy of Sciences, 118(6) – Published 9 February, 2021
DOI: 10.1073/pnas.2018379118