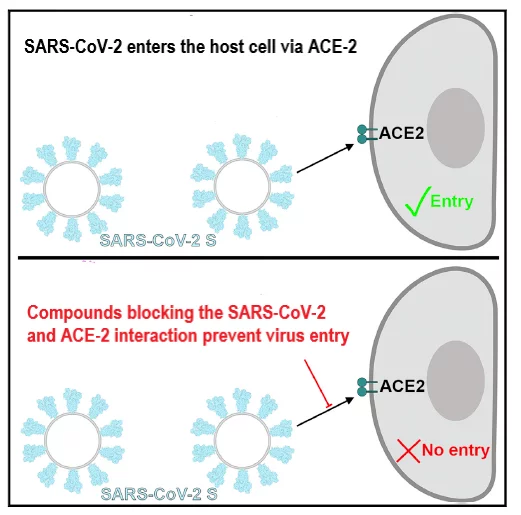
The spike glypcoprotein S of SARS-CoV-2 uses the human Angiotensin Converting Enzyme 2 (ACE2) as an entry receptor and recognizes it with a similar affinity to the 2002–2003 SARS-CoV isolates, which suggests it can spread efficiently in humans, in agreement with the numerous SARS-CoV-2 human-to-human transmission events reported to date (Walls et al., 2020).
In collaboration with its academic partners at the PSI, InterAx Biotech is establishing a pseudovirus entry assay using murine leukemia virus based pseudotyped particles expressing the SARS-CoV-2 spike protein (and other relevant SARS-CoV-2 envelope proteins). The assay is described in detail in Walls et al. (2020) and Millet et al. (2016, 2019) and will enable a medium-throughput (96 well format) screen for blocking the interaction of the SARS-CoV-2 spike glycoprotein (and other envelope proteins) and the human ACE2 expressed in a host target cell line. This assay is suitable to be performed in the BSL-2 facilities at PSI and non-infective compounds ranging from small molecules to antibody sera can be tested. For detailed information please send an Email to contact@interaxbiotech.com with COVID-19 in the header.
References
Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell. 2020 Mar 6. pii: S0092-8674(20)30262-2.
Millet JK, Tang T, Nathan L, Jaimes JA, Hsu HL, Daniel S, Whittaker GR. Production of Pseudotyped Particles to Study Highly Pathogenic Coronaviruses in a Biosafety Level 2 Setting. J Vis Exp., 2019 Mar 1;(145).
Millet JK, Whittaker GR. Murine Leukemia Virus (MLV)-based Coronavirus Spike-pseudotyped Particle Production and Infection. Bio Protoc. 2016 Dec 5;6(23). pii: e2035
Contact
Department of Biology and Chemistry
Paul Scherrer Institute PSI
5232 Villigen-PSI
Switzerland
Scientist
Laboratory of Nanoscale Biology
Dr. Roger Benoit