The Center for Radiopharmaceutical Sciences (CRS) focuses on the development of new radiopharmaceuticals for positron emission tomography (PET) and other applications in nuclear medicine. Researchers at CRS have previously developed an 18F-based PET tracer (18F-AzaFol) for targeting the folate receptor, which is expressed on tumor cells, but also on activated macrophages involved in inflammatory processes. In a recent clinical study, 18F-AzaFol was investigated regarding its in vivo properties for folate-receptor targeting and assessed with respect to its safety profile in patients (NCT03242993).
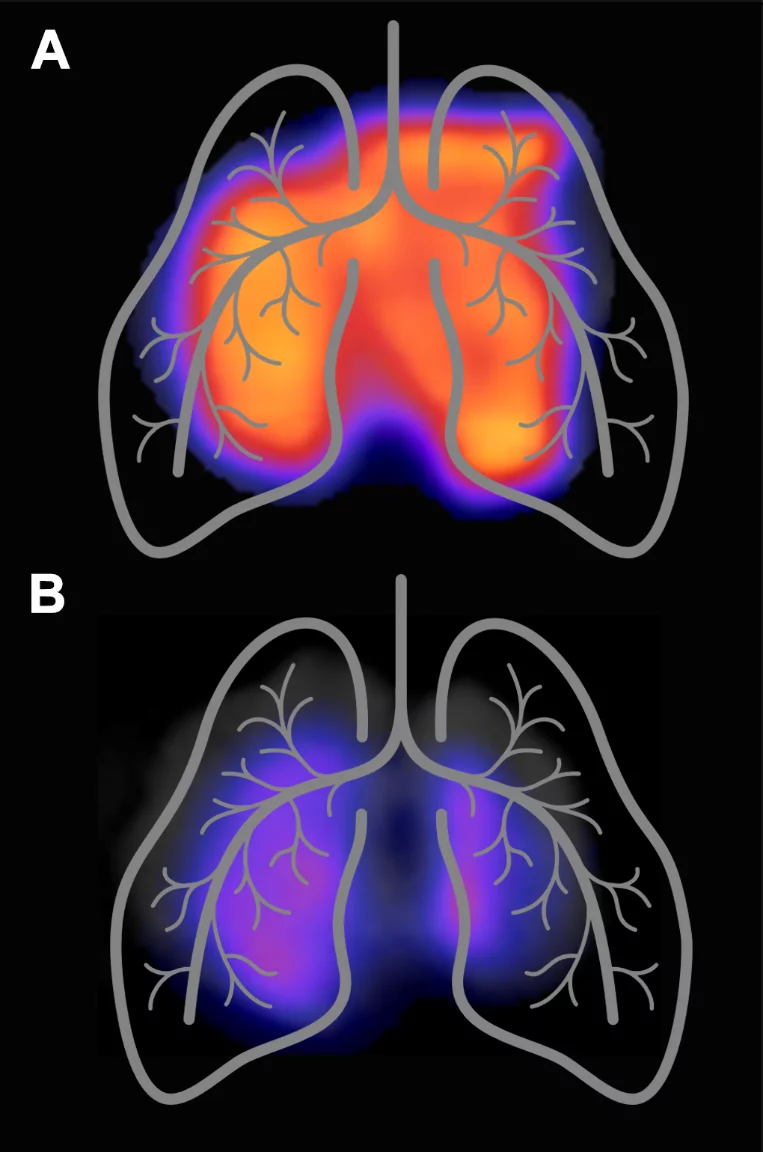
In an interdisciplinary project of PSI and the University Hospital Zurich, preclinical studies were performed to demonstrate the potential of 18F-AzaFol for imaging macrophages in lung inflammation. 18F-AzaFol was shown to accumulate specifically in diseased lung tissue but not in lungs of healthy mice. The increased abundance of folate receptor-expressing macrophages was confirmed on diseased murine and human lung tissue using several in vitro methods (Schniering et al, 2019 Front Immunol).
Alveolar macrophages play an important role during coronavirus infections and replication and are involved in antiviral response and defense mechanisms. Most importantly, acute respiratory distress syndrome, a complication that develops in approximately 20% of patients, results from exuberant host inflammatory responses with macrophages as key players. 18F-AzaFol/PET based macrophage imaging may thus, be useful to investigate the complex immune response to COVID-19 infection and possibly enable the development of personalized treatment regimens in the future.
In this context, also other targets may be of interest for nuclear imaging. Recently, it was suggested that integrin may act as an alternative receptor for SARS-CoV-2 and could be implicated in its transmission and pathology. Clinically used RGD radiopharmaceuticals for integrin targeting may, therefore, be considered as effective imaging tools. In a proof-of-concept study, increased accumulation of an RGD radiopharmaceutical was demonstrated in a preclinical setting of lung inflammation by the same research team (Schniering et al. 2018 Ann Rheum Dis).
Schniering J., Benešová M., Brunner M., Haller S., Cohrs S., Frauenfelder T., Vrugt B., Feghali-Bostwick C.A., Schibli R., Distler O., Müller C., Maurer B. Visualisation of interstitial lung disease by molecular imaging of integrin αvβ3 and somatostatin receptor 2. Ann Rheum Dis (2018). (doi: 10.1136/annrheumdis-2018-214322)
Contact
Department of Biology and Chemistry
Paul Scherrer Institut
5232 Villigen-PSI
Switzerland
Scientist
Laboratory of Nanoscale Biology
Dr. Roger Benoit