In collaboration with research groups from PSI and Universities we are developing surfaces for application in biomolecular and biomaterials research ranging from protein patterns on wafer, glass and polymer surfaces to micro- and nanostructured surfaces and templates for replication in biocompatible polymers.
Specific projects
Polymer brush structures for biosensor applications
Based on our earlier work on polymer micro- and nanografting, we are exploring the potential of polymer brush structures for applications as bio-interfaces. In particular, we’re grafting copolymers consisting of hydrophilic units with antifouling properties and monomer that can easily biofunctionalized from the surface of polymer foils. Biofunctionalization is currently focused towards biosensor applications and includes binding of enzymes, linker proteins such as streptavidin and DNA.
Reference
M. Moazeni, P. Berger and C. Padeste, Polymer brush structures functionalized with molecular beacon for point-of-care diagnostics.
Micro and Nano Engineering. 2023; 19: 100184 (9 pp.). https://doi.org/10.1016/j.mne.2023.100184
Earlier Projects
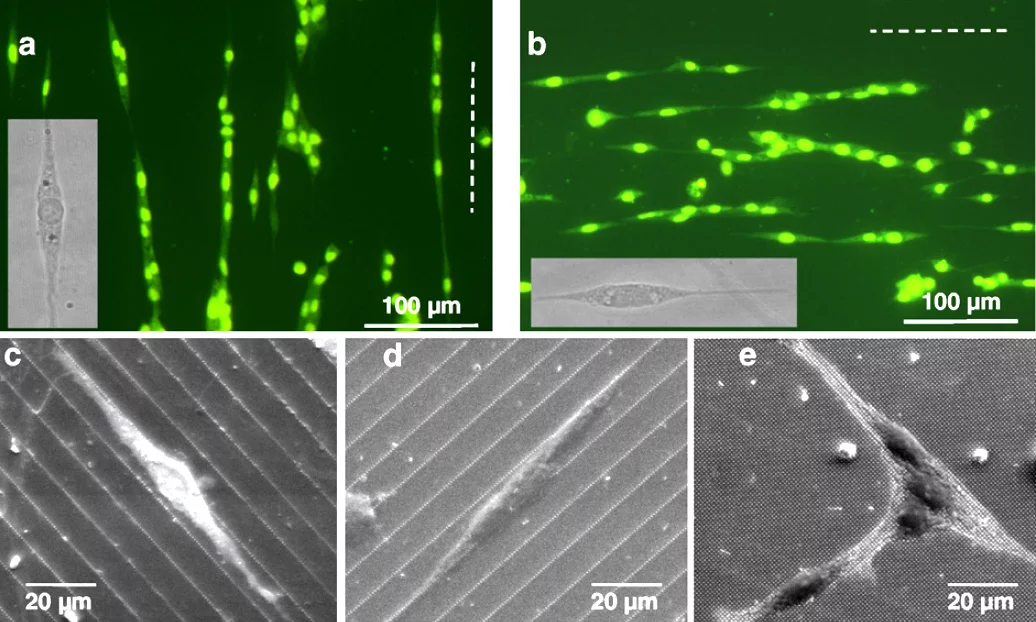
Substrates for ultrastructural studies We are developing patterned supports for neuron cell co-cultures with the aim to replicate the neuronal pathway under controlled conditions for subsequent use in ultrastructural studies of Parkinson’s Disease (PD). The lithographically structured substrates are compatible with a cryo-ultrastructural analysis workflow consisting of high pressure freezing, freeze substitution and preparation for electron microscopy. The aim is to understand the ultrastructure of PD-associated sub-cellular aggregates and intracellular components within diseased synapses, axons and neuronal bodies, in order to design better therapeutic strategies and ways to potentially monitor and detect the disease at an early stage. Our well-designed cell culture model can provide key insights while circumventing complex issues associated with handling whole human brain tissue.
Reference: Tran HT, Lucas MS, Ishikawa T, Shahmoradian SH and Padeste C (2021) A Compartmentalized Neuronal Cell-Culture Platform Compatible With Cryo-Fixation by High-Pressure Freezing for Ultrastructural Imaging. Front. Neurosci. 15:726763. doi: 10.3389/fnins.2021.726763
Integrated microneedle-optofluidic biosensor In an international collaboration between the University of the British Columbia (UBC) and the Paul Scherrer Institut (PSI), a promising system for painless and minimally-invasive therapeutic drug monitoring has been demonstrated. The proposed device is based on the combination of an optofluidic system with hollow microneedles to extract extremely small volumes (< 1 nL) of interstitial fluid (ISF) to measure drug concentrations. >>read more
VEGF-Patterns Based on photolithograpghy or on microfluidics we were producing patterns of proteins of the VEGF-Family. Studies of porcine aortic endothelial cells (PAE cells) cultured on the substrates enable new insights into the mechanisms deteriming blood vessel formation which are highly dependent on the presence and distribution of VEGF-proteins. In collaboration with Kurt Ballmer, molecular cell biology at PSI.