The vanadium redox flow battery (VRFB) is designed for grid-scale energy storage applications. Ion-exchange membranes are performance and cost relevant components of redox flow batteries. Currently used materials are largely ‘borrowed’ from other applications that have different functional requirements. For next generation VRFBs, it would be desirable to develop membrane materials based on low-cost porous separators with low resistance and high transport selectivity to minimize vanadium-ion and electrolyte crossover.
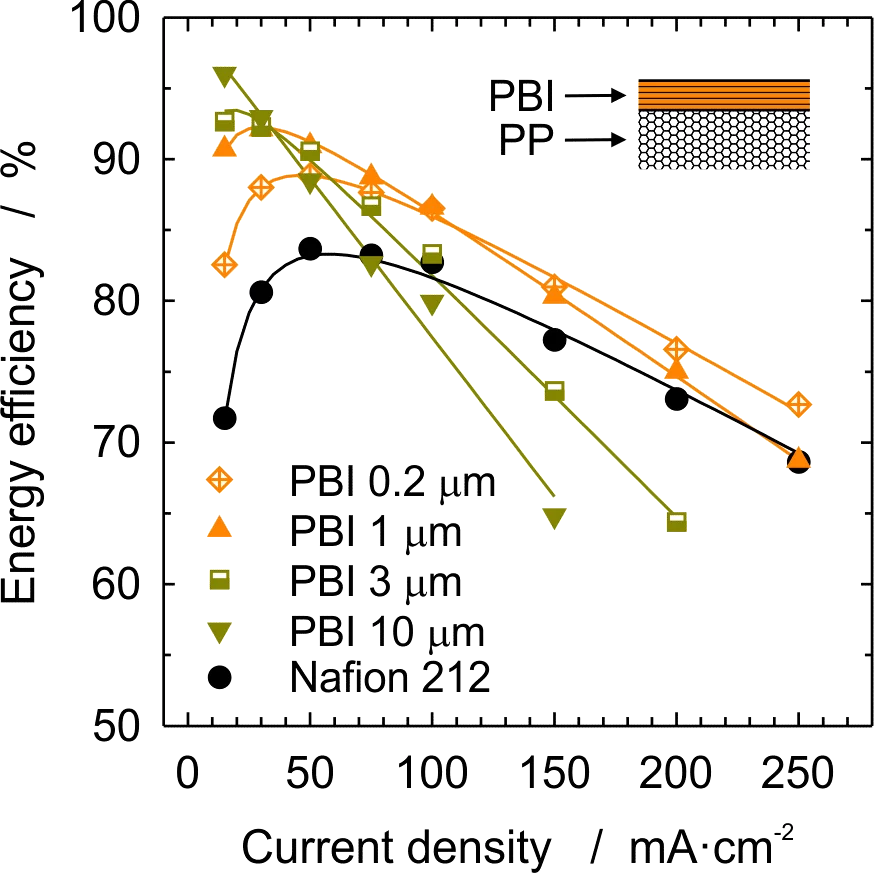
We present a composite membrane for the VRFB consisting of a porous propylene (PP) separator of 30 µm thickness laminated with a thin meta-polybenzimidazole (mPBI) layer. PBI films are prepared by solution casting to obtain thicknesses in the range of 0.2 to 10 µm. Upon immersion of PBI in the electrolyte solution containing 1.6 M vanadium in 2 M sulfuric acid, the benzimidazole units of PBI are protonated, hence creating an ion conducting polymer. Ohmic resistance of PBI equilibrated in the electrolyte was measured at ~30 mΩ·cm2 per micron of film thickness. This offers the prospect of designing a composite membrane-separator with low ohmic resistance. PP-PBI composite membranes with PBI layer thickness ranging from 0.2 to 10 µm were tested in single laboratory cells and compared to a cell with Nafion 212 membrane. For all PBI thicknesses, an improved coulombic efficiency was measured. The voltage efficiency was similar to that with Nafion 212 for the membrane with the thinnest PBI layer (0.2 µm), thicker PBI layers induced increasing ohmic losses. The overall charge-discharge efficiency was superior to that with Nafion 212 for the membranes with 0.2 and 1 µm PBI thickness up to a current density of 0.2 A/cm2. These results demonstrate that with a careful choice of constituents and architecture, it is possible to prepare membranes with high performance, high efficiency and low cost for next generation redox flow battery systems.
Contact
PD Dr. Lorenz Gubler
Head Membranes & Electrochemical Cells Group
Paul Scherrer Institut
5232 Villligen PSI
Telephone: +41 56 310 26 73
E-mail: lorenz.gubler@psi.ch
Original Publication
Composite Membranes Containing a Porous Separator and a Polybenzimidazole Thin Film for Vanadium Redox Flow Batteries
Lorenz Gubler, David Vonlanthen, Aaron Schneider, Fabio J. Oldenburg
Journal of The Electrochemical Society
DOI: 10.1149/1945-7111/ab945f
Acknowledgement
Swiss National Science Foundation and Innosuisse: Bridge Discovery program (grant number 40B2–0_176653).