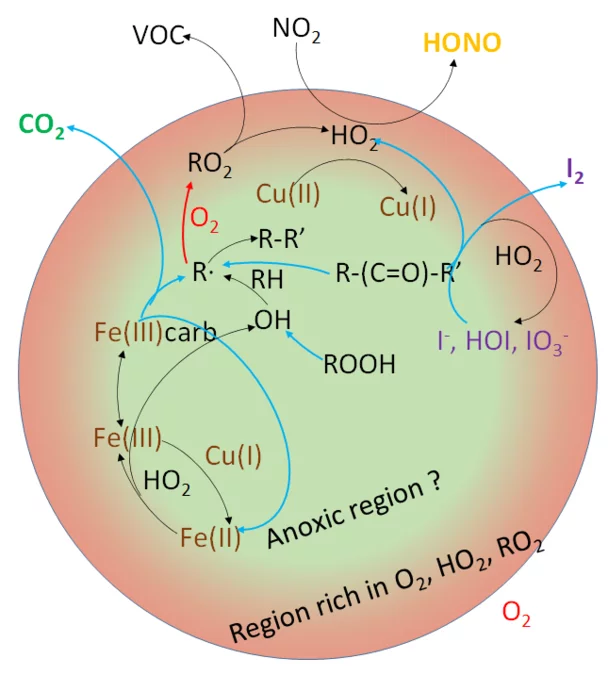
Reactive oxygen species (ROS) such as hydroxyl radicals, hydrogen peroxide, superoxide and others are formed by reduction of oxygen in radical cycles initiated by photolysis of inorganic and organic precursors typically present in atmospheric aerosol particles. These species contribute to the oxidative processing of aerosol particles, especially to its secondary organic components that derive from biogenic or anthropogenic volatile organic compounds (VOC). ROS are involved in redox processes of nitrogen and halogen compounds that feed back to gas phase composition. ROS are also suspected to be at the origin of health effects of aerosol via inducing oxidative stress upon inhalation. Multiphase processing in aerosol particles is strongly affected by condensed phase diffusion that may limit reaction rates and the exchange with the gas phase. The condensed phase (aqueous solution) - air interface is of particular importance as it offers a chemical and physical environment distinctly different from the aqueous bulk in terms of composition, hydration state of molecules and surface specific reaction route. Following up from the previous projects, we use photolysis of carbonyls, transition metal complexes and peroxides to initiate radical chemistry and then study the impact of Fe and Cu ions and other redox active species on the fate of nitrogen oxide and halogen compounds, on ROS production and on secondary organic aerosol proxies. For the nitrogen oxides, we will specifically look at the involvement of ROS in the formation of nitrous acid (HONO), an important radical precursor in the troposphere. For halogens, the focus will be on cycling of iodine species and release of molecular iodine to the gas phase. We will specifically address the limitation of diffusion of molecular oxygen and peroxy radicals and the concomitantly occurring anoxic conditions in particle interiors that will be probed by the nitrogen and halogen species, by organic radical recombination products, by the total ROS production and by directly and in situ probing chemical gradients within particles. This will be accomplished by a combination of the surface sensitive X-ray photoelectron spectroscopy, online extractive electro-spray ionisation aerosol mass spectrometry and scanning transmission X-ray spectromicroscopy. Experiments will be accompanied by explicit aqueous phase chemical modelling using the CAPRAM model and by process modelling with a depth-resolving layer model. Implications will be assessed by box models and larger scale models, in collaboration with external partners.
Funding
Swiss National Science Foundation: 188662
Team
Selected Publications
-
Corral Arroyo P, David G, Alpert PA, Parmentier EA, Ammann M, Signorell R
Amplification of light within aerosol particles accelerates in-particle photochemistry
Science. 2022; 376(6590): 293-296. https://doi.org/10.1126/science.abm7915
DORA PSI -
Alpert PA, Dou J, Corral Arroyo P, Schneider F, Xto J, Luo B, et al.
Photolytic radical persistence due to anoxia in viscous aerosol particles
Nature Communications. 2021; 12(1): 1769 (8 pp.). https://doi.org/10.1038/s41467-021-21913-x
DORA PSI -
Gladich I, Chen S, Yang H, Boucly A, Winter B, van Bokhoven JA, et al.
Liquid-gas interface of iron aqueous solutions and Fenton reagents
Journal of Physical Chemistry Letters. 2022; 13(13): 2994-3001. https://doi.org/10.1021/acs.jpclett.2c00380
DORA PSI -
Chen S, Artiglia L, Orlando F, Edebeli J, Kong X, Yang H, et al.
Impact of tetrabutylammonium on the oxidation of bromide by ozone
ACS Earth and Space Chemistry. 2021; 5(11): 3008-3021. https://doi.org/10.1021/acsearthspacechem.1c00233
DORA PSI -
Dou J, Alpert PA, Corral Arroyo P, Luo B, Schneider F, Xto J, et al.
Photochemical degradation of iron(III) citrate/citric acid aerosol quantified with the combination of three complementary experimental techniques and a kinetic process model
Atmospheric Chemistry and Physics. 2021; 21(1): 315-338. https://doi.org/10.5194/acp-21-315-2021
DORA PSI -
Dou J, Luo B, Peter T, Alpert PA, Corral Arroyo P, Ammann M, et al.
Carbon dioxide diffusivity in single, levitated organic aerosol particles
Journal of Physical Chemistry Letters. 2019; 10(15): 4484-4489. https://doi.org/10.1021/acs.jpclett.9b01389
DORA PSI