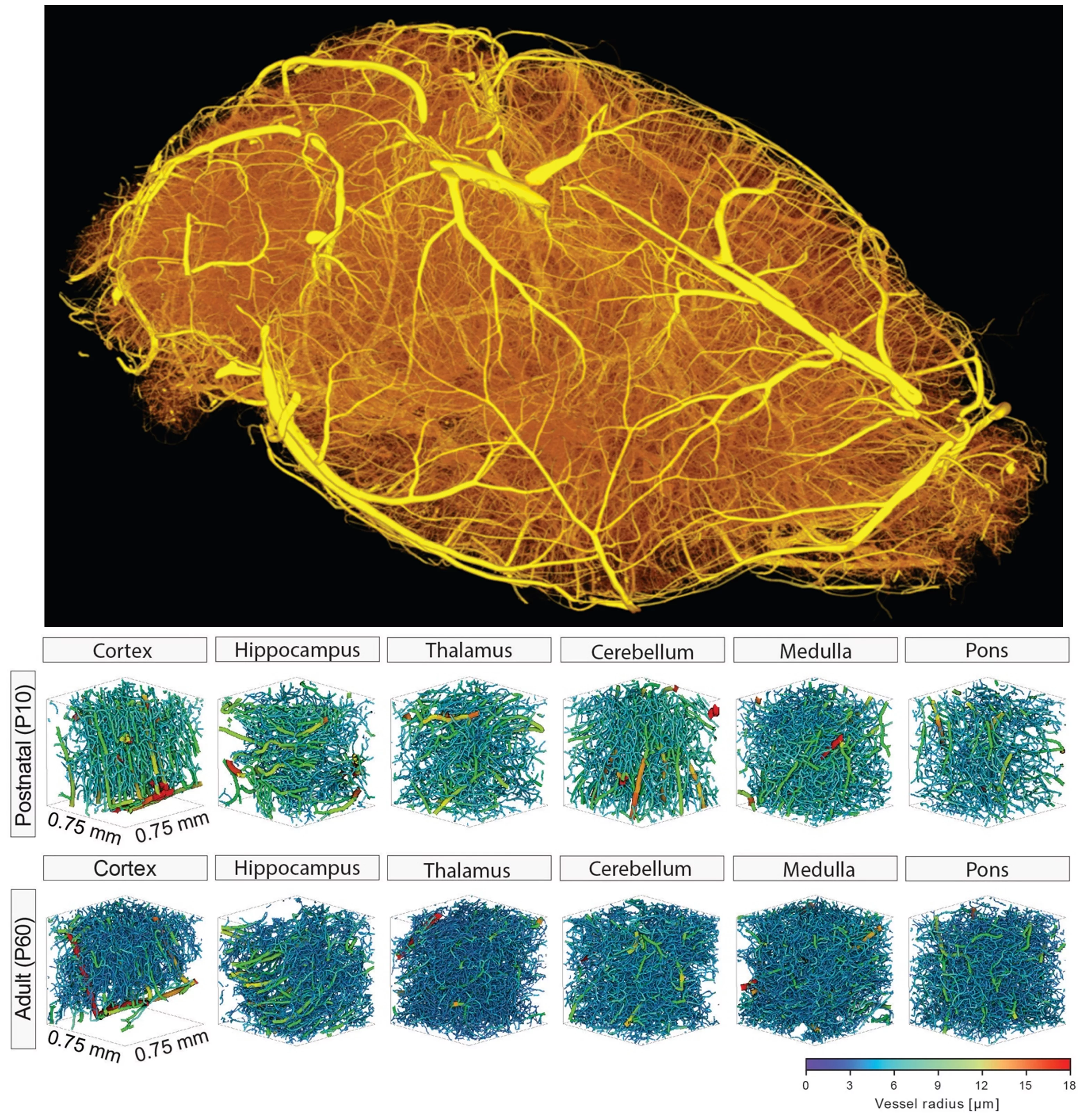
The human brain crucially depends on its vasculature for proper functioning as despite accounting for only 2% of the body mass it consumes 20% of the cardiac output and 20% of the oxygen and glucose of the entire body, corresponding to a 10-fold higher rate of energy metabolism compared to an equivalent tissue volume in the rest of the body. The brain vasculature is constituted of a complex 3D vascular network of an astonishing 644 kilometers (400 miles), anatomically organized so that each individual neuron is not more than 22 to 27 mm (in humans, depending on the cortical depth) away from the closest microcapillary, thereby assuring proper oxygen delivery. In the mouse brain, the absolute numbers are obviously lower but show similar relative values. Cerebral angiogenesis in mice and humans (and in all mammals) is highly active during embryonic and postnatal brain development and almost quiescent in the adult healthy brain, but is reactivated in vascular-dependent central nervous system pathologies.
A reliable method to address how the brain vasculature network shapes from the developmental to the adult stage is crucial for a better understanding of underlying vascular dynamics also in pathological conditions. In this work we developed a protocol that enables hierarchical imaging and computational analysis of vascular networks in entire postnatal- and adult mouse brains. This enabled direct and quantitative comparisons of the morphological brain vascular network architecture between different postnatal and / or adult developmental stages as well as to distinguish capillaries and non-capillary blood vessels along with various parameters such as vascular volume fraction, vascular branch point density and -degree, vessel segment length, -diameter, -volume, -tortuosity, -directionality, and extravascular distance, throughout the entire mouse brain vasculature.
The different stages of the procedure include resin-based vascular corrosion casting, scanning electron microscopy, synchrotron radiation- and desktop µCT imaging, combined with computational network analysis. The protocol allows the influence of any angiogenic molecule to be studied including pharmacological compounds and blocking antibodies, and to test their pro- or antiangiogenic effects on vascularization and various specific parameters of the vascular network in postnatal as well as adult wild-type or genetically modified mice. Detailed visualization and quantification of the three-dimensional brain vasculature can now be obtained with yet unmet precision and this approach can be used to establish a detailed morphological atlas of the entire mouse brain vasculature at both the postnatal and the adult stage of development, in health, as well as disease.
Original Publication
Wälchli, T., Bisschop, J., Miettinen, A. et al. Hierarchical imaging and computational analysis of three-dimensional vascular network architecture in the entire postnatal and adult mouse brain. Nat Protoc (2021).
Contact
Forschungsstrasse 111
CH-5232 Villigen