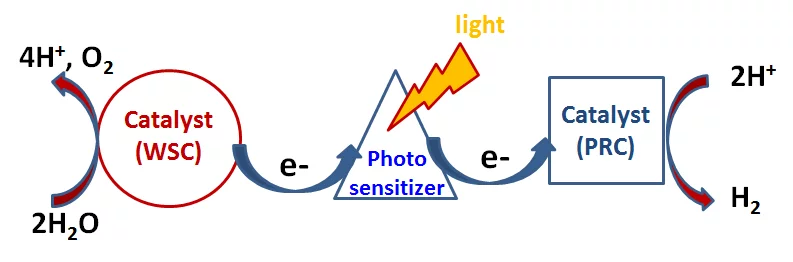
A technology to use the energy of sunlight to produce fuels is highly desirable for replacing fossil fuels with a renew-able energy source. The promising approach for achieving this goal is to mimic photo-synthesis and develop molecular catalysts for water splitting and hydrogen evolution. This field is known as artificial photosynthesis. The most simple artificial photosynthesis process is splitting of water into molecular oxygen and hydrogen. It consists of two half-reactions – the oxidative O2 evolving reaction and reduction of protons generating H2. With time-resolved X-ray absorption spectroscopy we selectively look either at photosensitizer or on catalyst in the photocatalytic system. Since two half-reactions require different catalysts, we study them separately.
Review of our results in this field is available:
-
Smolentsev G, Sundström V
Time-resolved X-ray absorption spectroscopy for the study of molecular systems relevant for artificial photosynthesis
Coordination Chemistry Reviews. 2015; 304-305: 117-132. https://doi.org/10.1016/j.ccr.2015.03.001
DORA PSI
Hydrogen evolving molecular catalysts
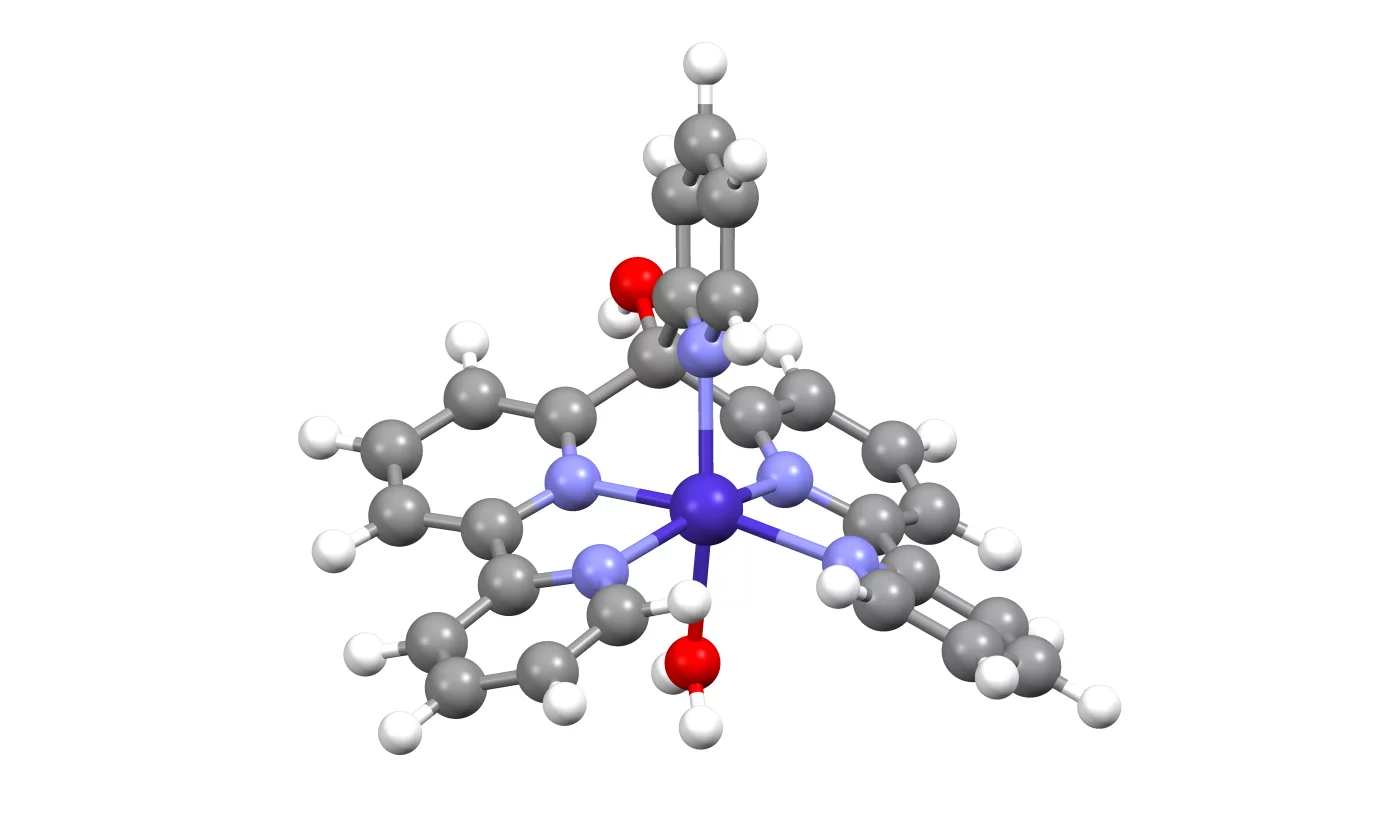
The focus of this project is on molecular catalysts with single 3d metal center, in particular Co. The goal is to catch Co(I), Co(III)H and other relevant intermediates of hydrogen evolution reaction which are formed in the time range from hundreds of nanoseconds to seconds. We determine the structure of such intermediates based on quantitative analysis of time-resolved XANES spectra, for example using FitIt code. Example of such application can be found in
-
Smolentsev G, Cecconi B, Guda A, Chavarot-Kerlidou M, van Bokhoven JA, Nachtegaal M, et al.
Microsecond X-ray absorption spectroscopy identification of CoI intermediates in cobaloxime-catalyzed hydrogen evolution
Chemistry: A European Journal. 2015; 21(43): 15158-15162. https://doi.org/10.1002/chem.201502900
DORA PSI
-
Smolentsev G, Soldatov MA, Probst B, Bachmann C, Azzaroli N, Alberto R, et al.
Structure of the CoI intermediate of a cobalt pentapyridyl catalyst for hydrogen evolution revealed by time-resolved X-ray spectroscopy
ChemSusChem. 2018; 11(18): 3087-3091. https://doi.org/10.1002/cssc.201801140
DORA PSI
Catalysts for water splitting
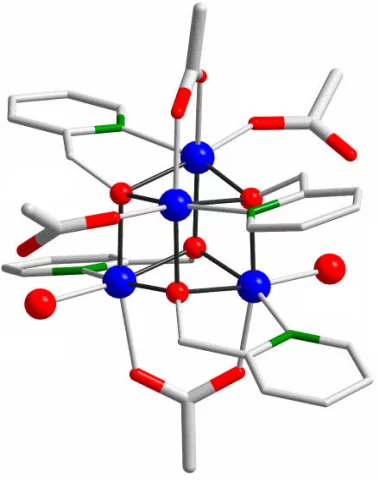
Within this project we investigate Co-based catalysts with cubane-type structure using time-resolved X-ray absorption spectroscopy. We have performed in-situ measurements of this catalyst during oxygen production reaction. The project is supported by NCCR MARVEL.
-
Song F, Moré R, Schilling M, Smolentsev G, Azzaroli N, Fox T, et al.
{CO4O4} and {CoxNi4-xO4} cubane water oxidation catalysts as surface cut-outs of cobalt oxides
Journal of the American Chemical Society. 2017; 139(40): 14198-14208. https://doi.org/10.1021/jacs.7b07361
DORA PSI