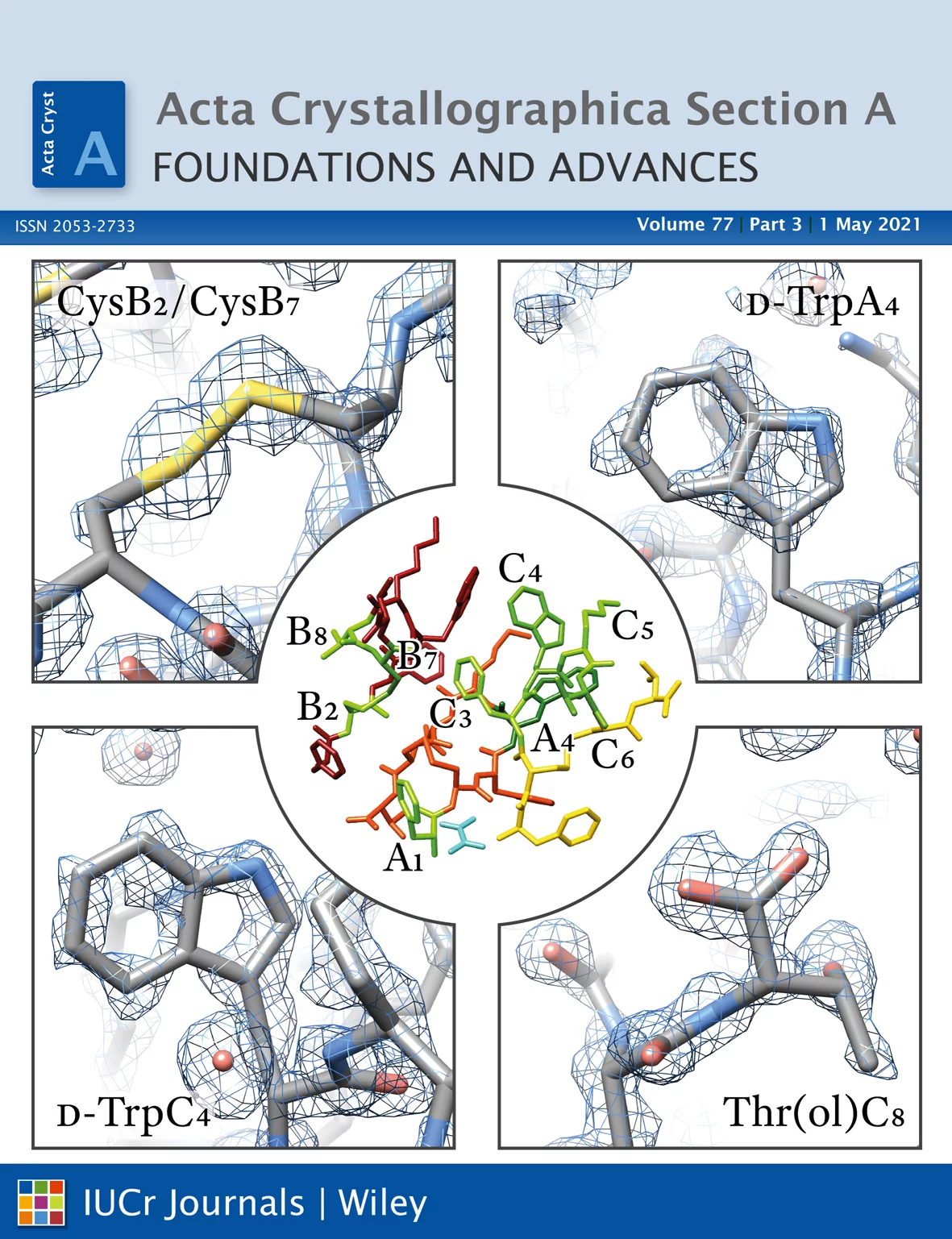
Using the methodology and the experimental protocols that Excelsus developed for the detection of traces of crystalline forms in pharmaceutical compounds, Excelsus team collected outstanding data on protein powders allowing the Biochemistry & Structural Biology team at the University of Patras to achieve a 1.8 Ang structural model of the molecule “octreotide”, surpassing the model the same group achieved in the past (see: earlier result.). This model corresponds to the highest resolution ever achieved for a peptide/ protein using X-ray powder diffraction and crystallographic methods. Furthermore, traces of minority crystalline impurities were detected for the first time. Structural model available here.
Advances in instrumentation, as well as the development of powerful crystallographic software have significantly facilitated the collection of high-resolution diffraction data and have made X-ray powder diffraction (XRPD) particularly useful for the extraction of structural information; this is true even for complex molecules, especially when combined with synchrotron radiation. In this study, in-line with past instrumental profile studies, an improved data collection strategy exploiting the MYTHEN II detector system together with significant beam focusing and tailored data collection options was introduced and optimized for protein samples at the Material Science beamline at the Swiss Light Source. Polycrystalline precipitates of octreotide, a somatostatin analog of particular pharmaceutical interest, were examined with this novel approach. XRPD experiments resulted in high angular and d-spacing (1.87 Å) resolution data, from which electron-density maps of enhanced quality were extracted, revealing the molecule's structural properties. Since microcrystalline precipitates represent a viable alternative for administration of therapeutic macromolecules, XRPD has been acknowledged as the most applicable tool for examining a wide spectrum of physicochemical properties of such materials and performing studies ranging from phase identification to complete structural characterization.
Fruitful discussions with the Materials Science beamline crew (Nicola Casati and Antonio Cervellino) and the PSI detector group (Bernd Schmitt and Anna Bergamaschi) have been instrumental to the success of the project.
Contact
Dr Fabia Gozzo, CEO
Excelsus Structural Solutions
PARK INNOVAARE, 5234 Villigen, Switzerland
16/13, Rue de la Cambre, 1200 Brussels, Belgium
+41 79 830 32 01
fabia.gozzo@excelsus2s.com
www.excelsusSS.com
Prof Irene Margiolaki
University of Patras
Department of Biology, Section of Genetics, Cell Biology and Development,
26500, Patras, Greece
+302610997408
imargiola@upatras.gr
https://sites.google.com/view/margiolaki-biology-upat/home?authuser=0
Original Publication
New perspectives in macromolecular powder diffraction using single photon counting strip detectors: High resolution structure of the pharmaceutical peptide, Octreotide; Maria Spiliopoulou, Fotini Karavassili, Dimitris-Panagiotis Triandafillidis, Alexandros Valmas, Christos Kosinas, Kleomenis Barlos, Kostas K. Barlos, Mickael Morin, Mathilde L. Reinle-Schmitt, Fabia Gozzo and Irene Margiolaki; Acta Cryst. (2021). A77, https://doi.org/10.1107/S2053273321001698