News
Biography
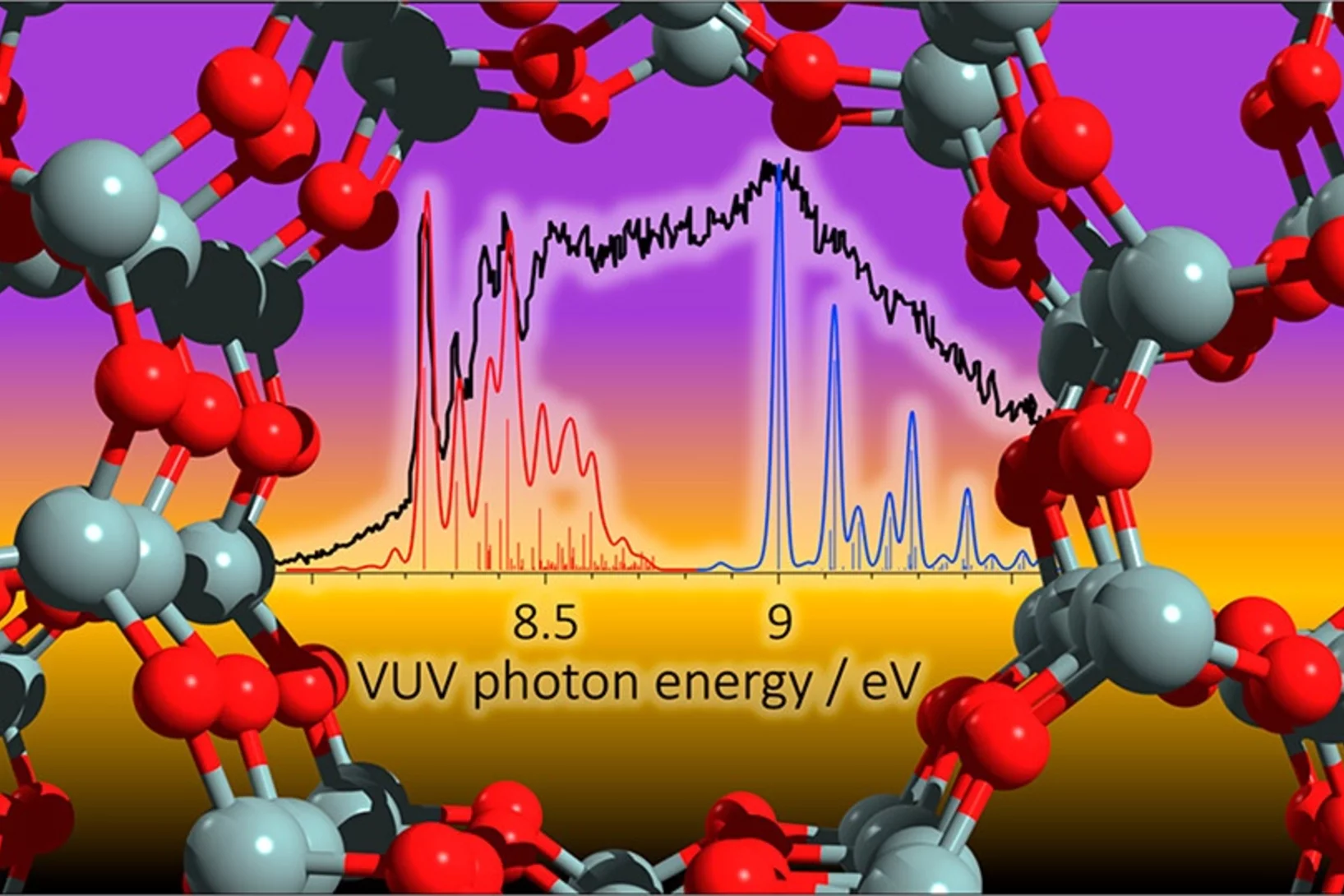
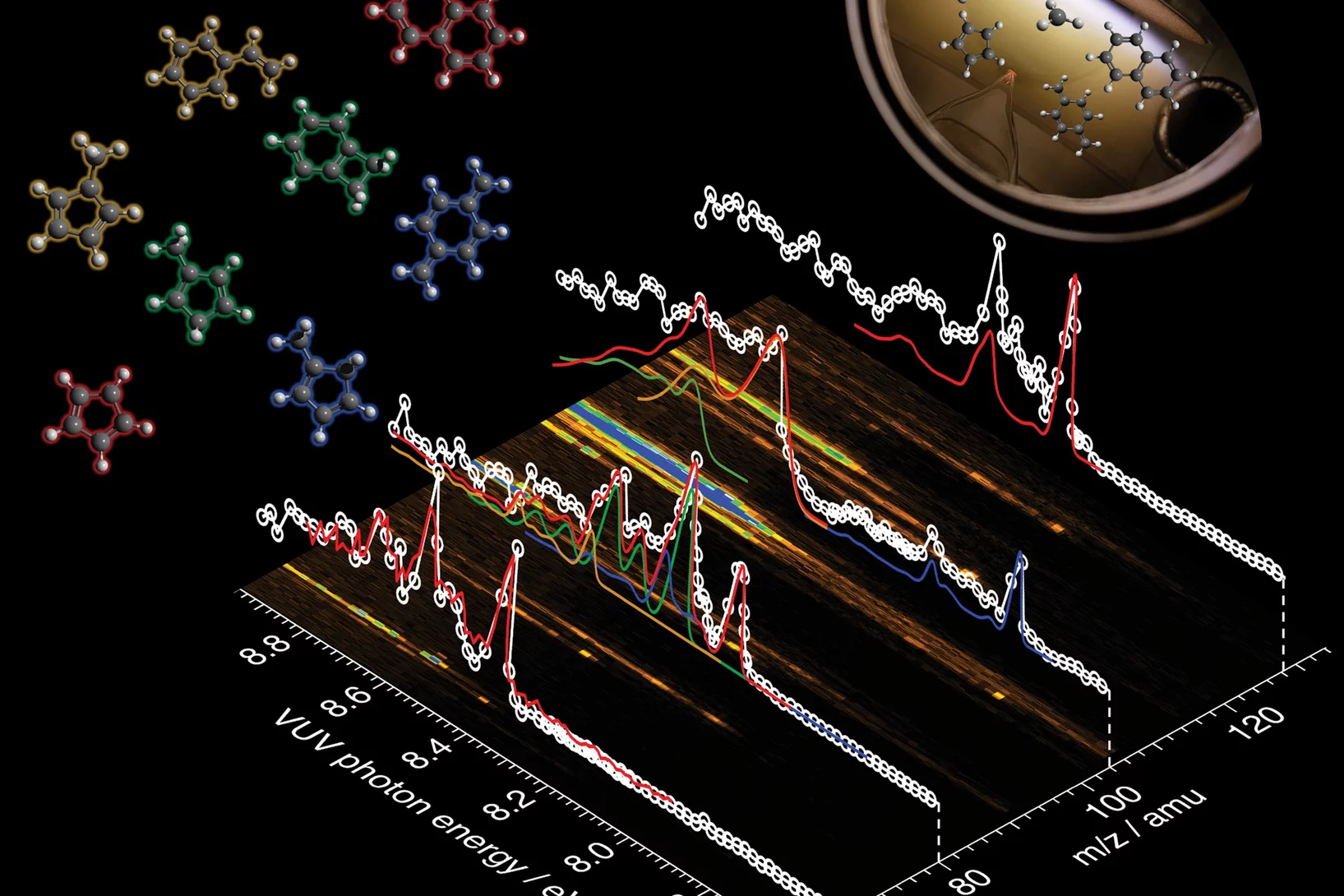
Patrick Hemberger is a principal investigator and beamline scientist at the vacuum ultraviolet (VUV) beamline at Paul Scherrer Institute. He obtained his PhD degree in physical chemistry from the University of Würzburg (Germany) with Ingo Fischer. Patrick develops and applies photoelectron photoion coincidence (PEPICO) techniques utilizing VUV synchrotron radiation to unveil reaction mechanisms at all states, time scales, and phases with a focus on heterogeneous catalysis. He received the Ruzǐcǩa Prize in Chemistry from ETH Zürich, was selected as a Mercator fellow by the German Science Foundation (DFG), and was recently recognized for his significant contributions to the research field of energy in the Rising Stars special issue of Energy and Fuels. Patrick has co-authored more than 200 publications in peer-reviewed journals. He was lecturer and local organizer of the Hercules School (2020-2022) in the field of Synchrotron Radiation and supervises PhD students and postdocs. Besides peer-reviewing activities for different journals and funding agencies he is a member of the Proposal Review Panel at Elettra Synchrotron, Trieste (Italy) and serves in the editorial advisory board of the Journal of Physical Chemistry (American Chemical Society).
Institutional Responsibilities
At PSI he is responsible for further developing and application of the technologies at the vacuum ultraviolet beamline for chemical reaction dynamics research. This includes development of sources for the generation of elusive and reactive intermediates (free radicals). With the ultimate goal to elucidate reaction mechanism in reactive flows, Patrick refines the instrumentation at the VUV beamline (spectrometers & sampling environment) to enable combination with novel reactors. He is heading the user chemistry laboratory at the Swiss Light Source, successfully applies for funding and is supervising PhD students and users.
Scientific Research
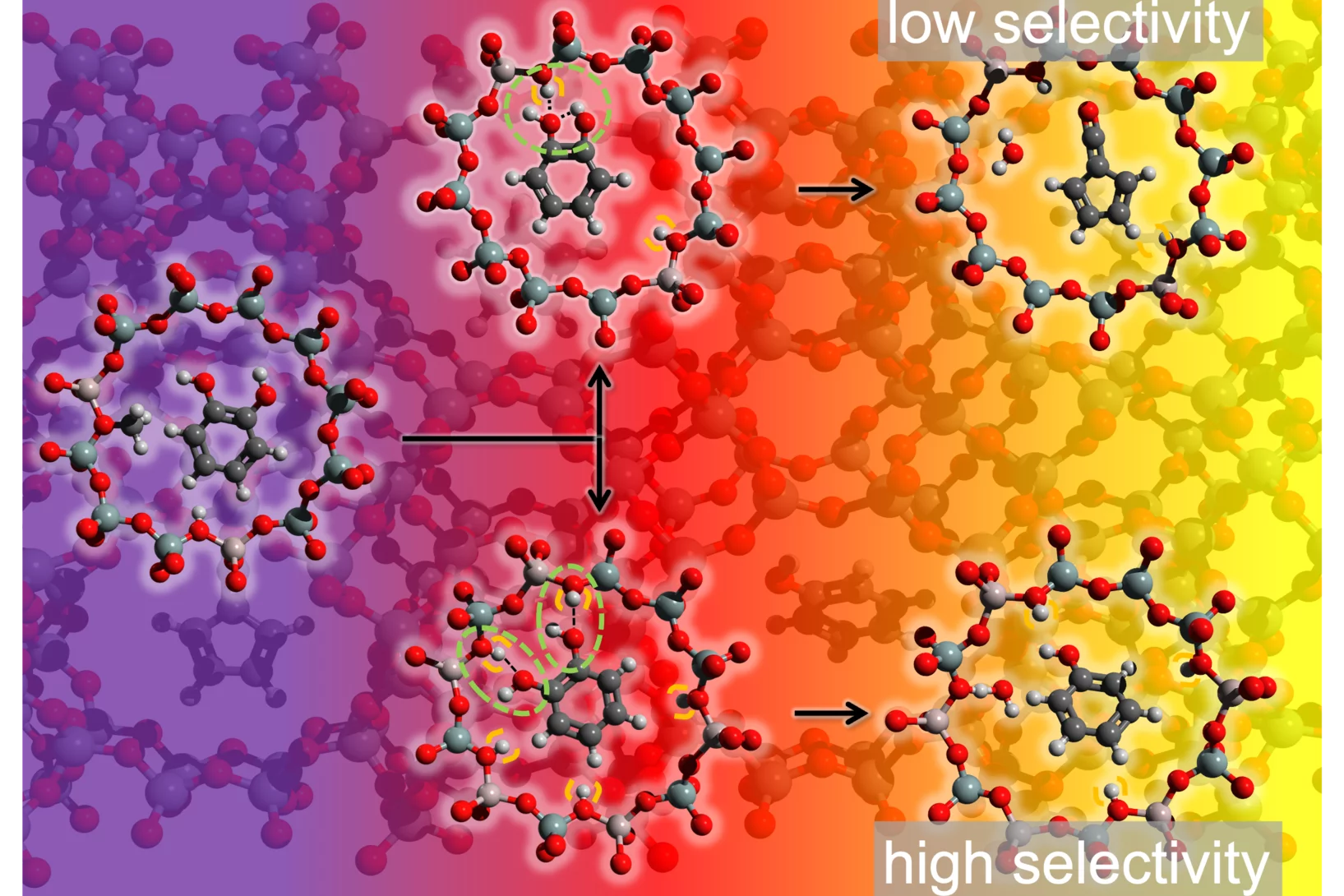
Patrick’s research interests are elucidating reaction mechanisms in reactive environments in all states, phases and time-scales. To unveil these insights, Patrick applies and develops methods to probe elusive and reactive molecules isomer-selectively utilizing vacuum ultraviolet synchrotron radiation, ultrafast lasers, photoelectron spectroscopy and mass spectrometric techniques. Currently he is principal investigator in several projects with the goal to elucidate reaction mechanisms at interfaces including heterogeneous catalytic processes as well as in processes involving the liquid phase. Besides projects with VUV synchrotron radiation, Patrick also applies time-resolved photoelectron spectroscopy and mass spectrometry to study the femtochemistry of free radicals.
Awards and Grants
- 2025/12: Appointment to Guest Research Professor at Shanghai Jiao Tong University.
- 2024/04: SNSF Grant: „Unveiling the Reaction Mechanism of Plastic Pyrolysis“, 398kCHF.
- 2022/10: Energy & Fuels Rising Star Award 2022.
- 2021: Finalist Falling Walls Breakthrough of the Year 2021 in the category Physical Sciences.
- 2021: NCCR Catalysis “Advanced Photoelectron Photoion Coincidence Spectroscopy to Unveil Reaction Mechanisms”, 240 kCHF
- 2020/11: Ružička Prize in Chemistry: "Understanding the mechanism of catalytic fast pyrolysis by unveiling reactive intermediates in heterogeneous catalysis"
- 2018/10: Cross Proposal: "Detecting reactive intermediates to understand the hydrothermal depolymerization mechanism of lignin", 540 kCHF
- 2018/04: SNSF Grant: "Disentangling the Reaction Mechanism in Catalytic Fast Pyrolysis of Lignin by Detecting Reactive Intermediates", 282 kCHF
- 2017: Poster Prize (Runner up) SCS Fall Meeting 2017 (Category: Catalysis Science & Engineering): "Understanding Reaction Mechanisms in Heterogeneously Catalyzed Reactions: The Case of Catalytic Fast Pyrolysis" link
- 2015: Grant: Swiss Federal Office of Energy (SFOE) Grant: "Disentangling soot formation processes applying VUV synchrotron radiation and ultrafast laser spectroscopy" 320 kCHF
- 2015: Mercator Fellow of the German Science Foundation "PEPICO in der Flammenforschung"
- 2012: SYN Budget Proposal for 2013, 100 kCHF
- 2008: Poster Prize Chem-SyStM 2008
Memberships
- Swiss Chemical Society SCS
- German Bunsen Society for Physical Chemistry
- German Chemical Society GDCh
- Editorial Advisory Board of Discover Catalysis (Springer)
- Editorial Advisory Board of the Journal of Physical Chemistry (American Chemical Society)