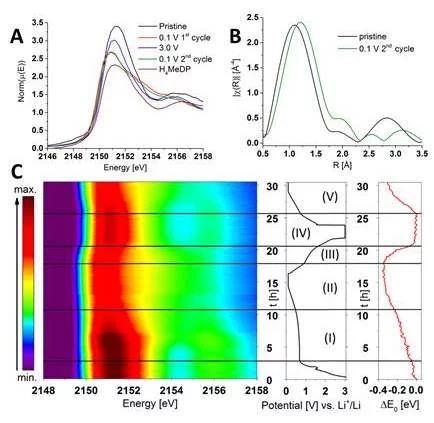
To date the electrochemical activity of battery materials was always relying in the oxidation/reduction of cationic redox (change of oxidation state of transition metals generally). However, recently, it was established in new cathode materials (so call Li-rich cathode) that the oxygen from the crystal lattice might also play the role of anionic redox center leading to enhance then the specific charge of battery materials. If the oxygen is now considered as anionic redox center, we wanted to know if other atoms could be considered also for this activity. When cycling diphosphonate-based organic-inorganic hybrid materials as negative battery electrodes, specific charges exceeding the maximum for a metal redox reaction are recorded. Classical explanations are electrolyte reduction/solid electrolyte interphase (SEI) oxidation. By an advanced operando X-ray absorption spectroscopy (XAS) at the P K-edge experiment, performed at the PHOENIX beamline, we demonstrate an additional contribution of reversible ligand co-cycling based on the P-atoms of the diphosphonate ligands, upon delithiation occurring during the first potential plateau. We thus investigate experimentally an anionic redox process in-situ so far not demonstrated in the literature. Thereby, we demonstrated that XANES is a method of choice to measure active materials under operando conditions, which could be applied to verify the predicted P co-cycling of other conversion-type negative electrode materials.
Contact
Prof. Dr. Petr NovakEnergy and Environment Division, OLGA 117
Paul Scherrer Institut
Telephone: +41 56 310 2457
E-mail: petr.novak@psi.ch
Dr. Claire Villevieille
E-mail: claire.villevieille@gmail.com
Dr. Thomas Huthwelker
Swiss Light Source
Paul Scherrer Institut
Telephone: +41 56 310 5314
E-mail: thomas.huthwelker@psi.ch
Original Publication
Phosphorus anionic redox activity revealed by operando P K-edge X-ray absorption spectroscopy on diphosphonate-based conversion materials in Li-ion batteriesSebastian Schmidt, Sebastien Sallard, Camelia Borca, Thomas Huthwelker, Petr Novak and Claire Villevieille
Chem. Commun., 2018, 54, 4939, 19 April 2018
DOI: 10.1039/c8cc01350k