Show filters
Gehörknöchelchen im Röntgenlicht – neue Technik enthüllt Strukturen in Rekordzeit
Am Beispiel eines Knochens demonstrieren PSI-Forschende, wie sich Strukturen biologischer Materialien von der Nano- bis zur Millimeterskala in kürzester Zeit erfassen lassen.
PILATUS4 detector arrives at PXIII
On April 4, 2025, a Dectris Pilatus4 2M detector was successfully installed at beamline PXIII. In the coming weeks, this new detector will be used to measure the first macromolecular crystallography (MX) experiments using the SLS 2.0 machine.
Thank You SLS
Our beamline scientists look back on 22 years of brilliant science made possible by the Swiss Light Source SLS.
Bright white coloring of Pacific cleaner shrimp revelead
In a study published in Nature Photonics, researchers at the Ben-Gurion University of the Negev, Israel, explain the bright, white-colored stripes from Pacific cleaner shrimps, one of the most efficient white reflectors found in nature.
Progress of the X06DA-PXIII beamline upgrade: First light in the optics hutch
On June 7, 2023, the PXIII project team successfully shone the first light into the optics hutch at the upgraded X06DA-PXIII beamline. It is an essential first step for testing new hardware and software solutions that will be implemented at SLS2.0.
SDU: Software for high throughput automated data collection at SLS MX
The Smart Digital User (SDU) software for unattended data collection has been deployed at the macromolecular crystallography beamlines at the Swiss Light Source.
A new spin on sample delivery for membrane proteins
Proteins hover in front of the X-ray beam at a Swiss Light Source beamline. Now, spinning thin films bring on board these trickiest of proteins.
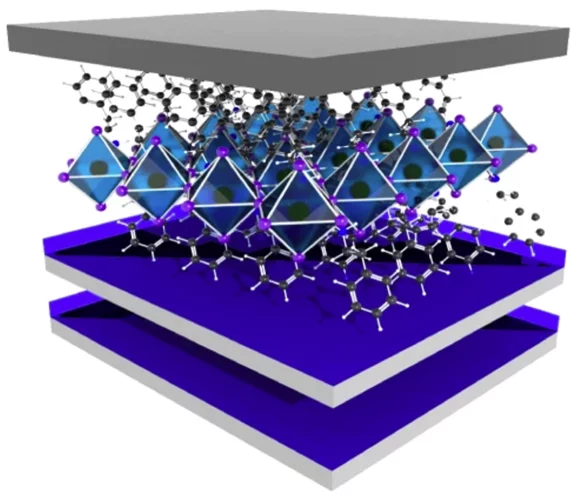
Lighting up the appealing world of hybrid perovskites
Researchers from Italy, in collaboration with the Paul Scherrer Institut, successfully used the macromolecular crystallography beamline X06DA-PXIII at the Swiss Light Source to characterize promising perovkites materials used in solar cells and other photodetector devices.
How to get chloride ions into the cell
A molecular movie shot at PSI reveals the mechanism of a light-driven chloride pump
Proteine auf Abstand
PSI-Forschende haben eine neue Methode entwickelt, um Proteine auf der Oberfläche von virusartigen Partikeln anzubringen.
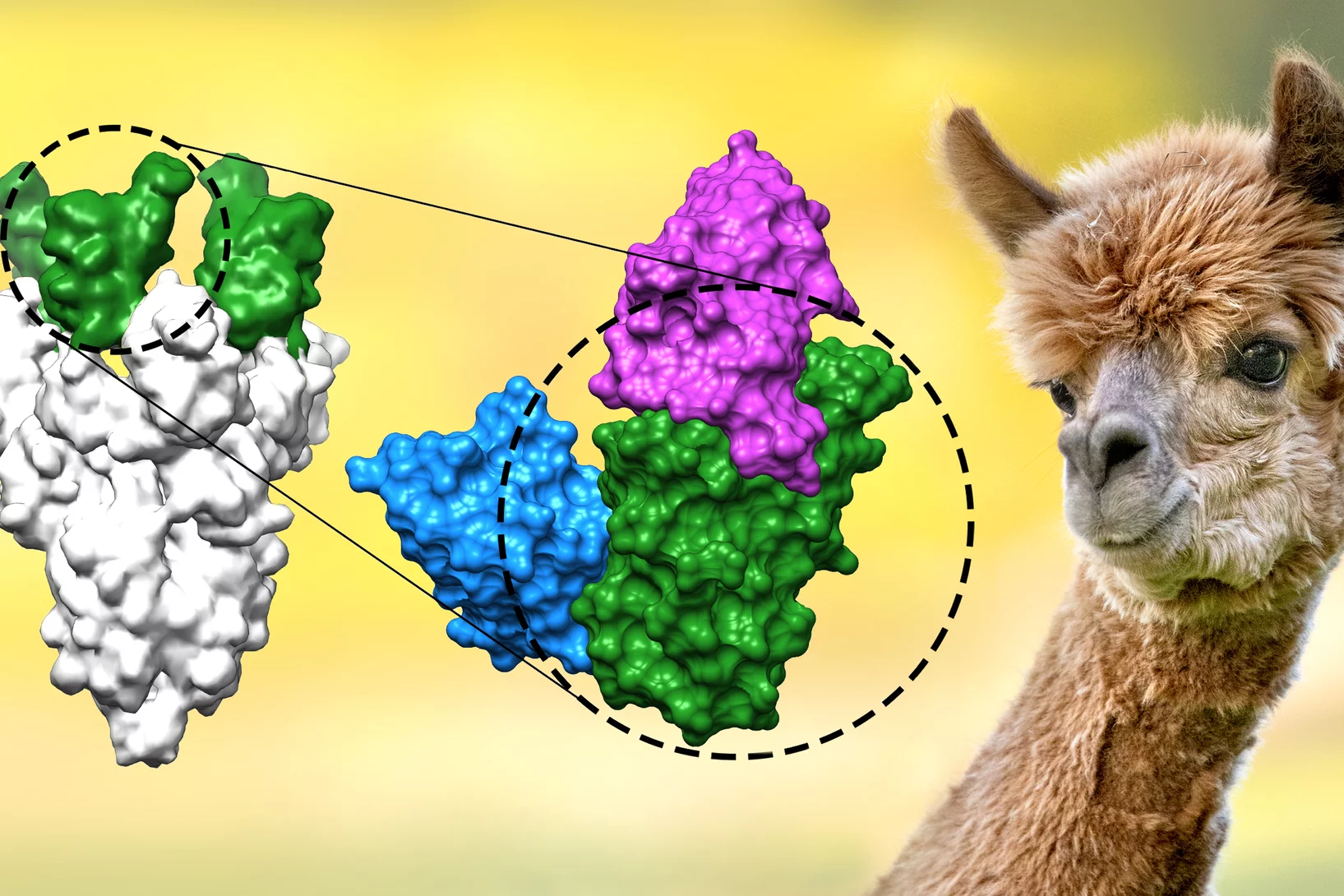
Nanobodies against SARS-CoV-2
In a study published in EMBO Journal, researchers at the Max Planck Institute for Biophysical Chemistry, Göttingen, Germany, developed nanobodies that efficiently block the coronavirus SARS-CoV-2 and its variants. The high resolution structural characterization was performed at the X10SA crystallography beamline at the Swiss Light Source.
How ethane-consuming archaea pick up their favorite dish
Scientists decode the structure of the enzyme responsible for the ethane fixation by – beside others – using the SLS.
On-demand sample delivery article highlighted in "Applied Physics Letters"
An article on the on-demand sample delivery and protein crystallography using acoustic levitation has been selected in an Applied Physics Letters collection of papers on technology and application of acoustic tweezers.
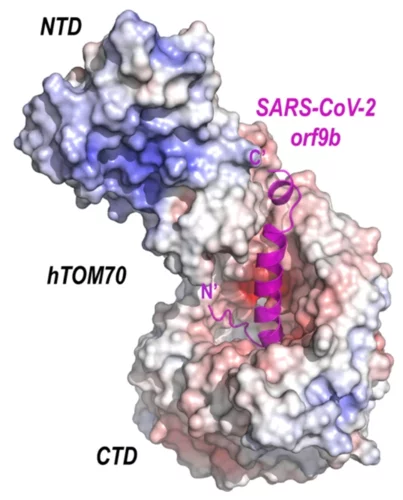
Crystal structure of SARS-CoV-2 Orf9b in complex with human TOM70 suggests unusual virus-host interactions
In a study published in Nature Communications, researchers at the NHC Key Laboratory of Systems Biology of Pathogens in Beijing, China, in collaboration with the Paul Scherrer Institut characterize the interactions of SARS-CoV-2 orf9b and human TOM70 biochemically, and they determine the 2.2 Å crystal structure of the TOM70 cytosolic domain with a bound SARS-CoV-2 orf9b peptide.
Wie Remdesivir gegen das Coronavirus wirkt
Forschende der Goethe-Universität Frankfurt haben in Kooperation mit dem PSI vermutlich einen weiteren, bislang unbekannten Wirkmechanismus des Virostatikums Remdesivir entdeckt.
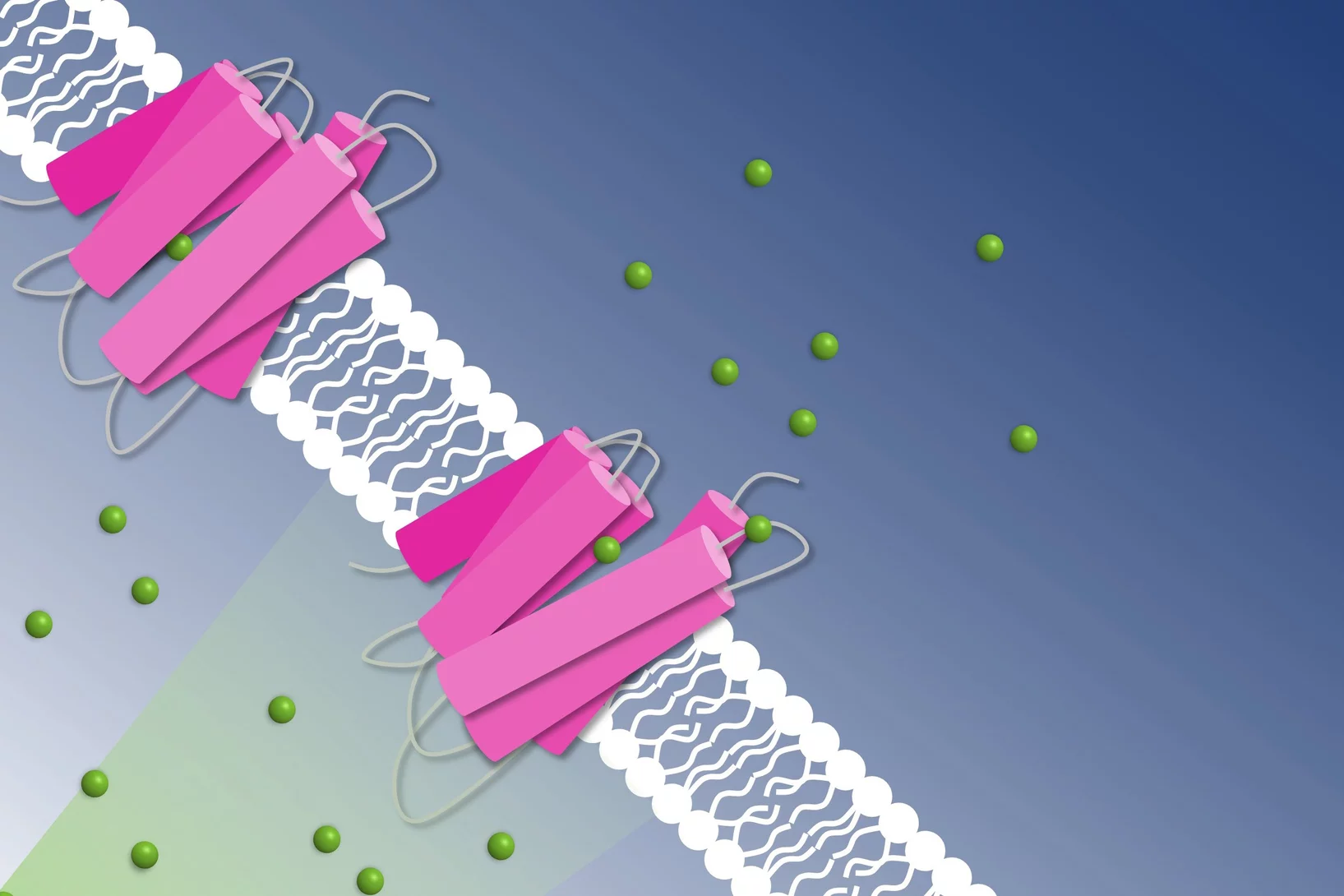
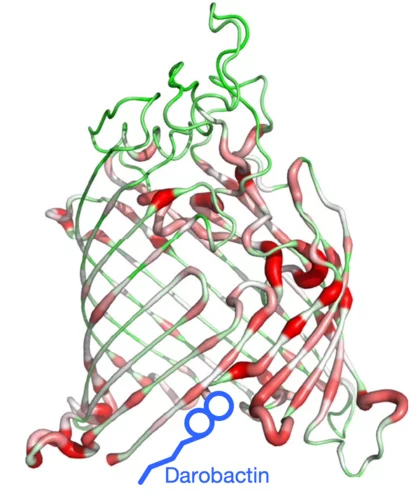
Combating antimicrobial resistance
In a study addressing the global health threat of drug resistance, researchers at the Biozentrum, University of Basel, have revealed how a new antibiotic, Darobactin, binds to the external membrane of gram-negative bacteria.
Forschung zu Covid-19 am Paul Scherrer Institut
Während viele Bereiche des Lebens eingeschränkt sind, bleiben wichtige Forschungsanlagen am PSI in Betrieb.
Abwarten und Kristalle züchten
Am PSI entschlüsseln Forschende die Struktur der Proteine von Bakterien und Viren. Mit diesem Wissen lassen sich beispielsweise Medikamente gegen Infektionskrankheiten entwickeln. Doch zuallererst muss ein äusserst kniffliges Problem gelöst werden: die Kristallisation der Moleküle.
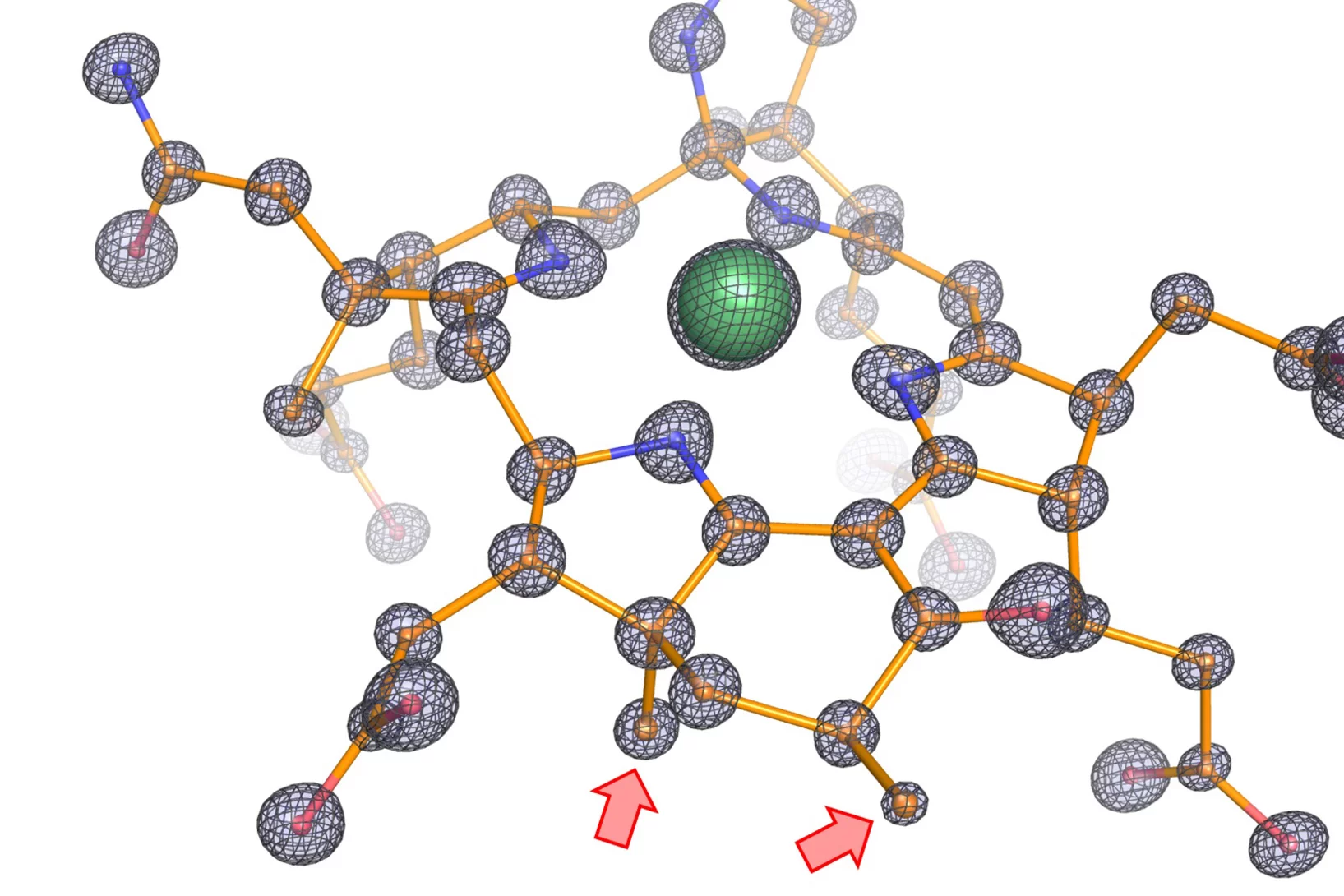
Eine Frage der Bindung
Am PSI screenen Forschende Molekülfragmente darauf, ob diese an wichtige Proteine des Coronavirus SARS-CoV-2 binden und dieses so möglicherweise lahmlegen können. Aus den vielen Einzelinformationen erhoffen sie sich eine Antwort darauf, wie ein wirkungsvolles Medikament aussehen kann.
Covid-19-Forschung: Antivirale Strategie mit Doppelwirkung
Frankfurter Wissenschaftler identifizieren eine mögliche Schwachstelle des SARS-CoV-2-Virus. Einen Teil ihrer Messungen führten sie an der Synchrotron Lichtquelle Schweiz SLS am PSI durch. Die Forschungsergebnisse erscheinen diese Woche im Fachblatt Nature.
First MX results of the priority COVID-19 call
The Dikic group at the Goethe University in Frankfurt am Main, Germany has published the first results following the opening of the "PRIORITY COVID-19 Call” at SLS.
SLS MX beamtime update
Update of the SLS MX beamline operation during the COVID-19 period
A crystal-clear picture
Fast and accurate data collection for macromolecular crystallography using the JUNGFRAU detector.
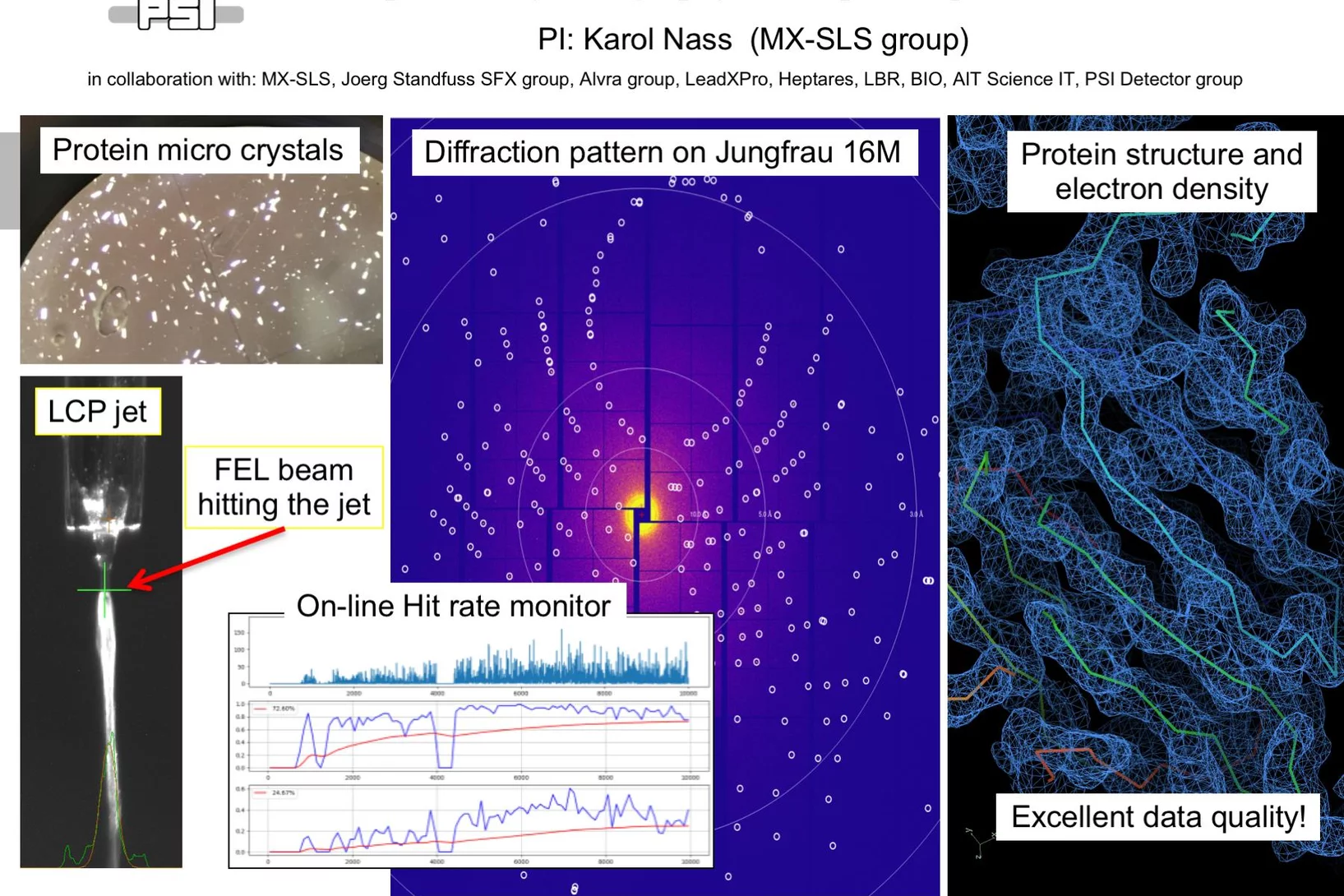
First serial femtosecond crystallography (SFX) pilot user experiment at SwissFEL
On the 7th to 12th of August 2018, a collaborative group of scientists from the Paul Scherrer Institute and members of the LeadXpro and Heptares pharmaceutical companies led by Karol Nass (PSI macromolecular crystallography MX-SLS group) performed the first serial femtosecond crystallography (SFX) pilot user experiment at the SwissFEL X-ray free electron laser (XFEL).
Rhine-Knee Regiomeeting 2018
Since it was first established in 1987, the annual Regio-Meeting has been instrumental in facilitating interactions in the structural biology community in southwestern Germany, the eastern region of France and an expanding area of Switzerland. It is set as an informal event to foster young scientists to discuss their research results in an international context. The 2018 edition will take place in the heart of Switzerland in Emmetten from September 26 to 28, 2018. Registration and abstract submission deadline: September 7, 2018.
In Situ Serial Crystallography 2 Workshop at the SLS
A workshop dedicated to the presentation of the in meso in situ serial crystallography (IMISX) method (Huang et al. 2015, 2016 ActaD) for the X-ray structure determination of membrane proteins is organised at the Swiss Light Source at PSI for the second time. It will be held between November 27 and 29, 2017.
Towards understanding of human betacoronavirus HKU1 life cycle
Researchers from China and USA join forces with Swiss Light Source (SLS) macromolecular crystallography (MX) beamline scientists in a study, which aims at understanding an important step in the life cycle of the human betacoronavirus HKU1.
1000 Structures solved at X06DA-PXIII
The macromolecular crystallography beamline X06DA-PXIII has reached 1,000 structures in the Protein Data Bank (PDB) on February 22, 2017.
First protein structure solved using the JUNGFRAU detector!
JUNGFRAU is a charge-integrating, two-dimensional pixel detector developed at the Paul Scherrer Institut for use at free-electron lasers, in particular SwissFEL, and synchrotron light sources. On the 10th October, the first protein crystallography experiment using the JUNGFRAU detector, was performed at the beamline X06SA (PXI) of the Swiss Light Source by the members of the Protein Crystallography and Detectors groups at PSI.
Call for expressions of interest: Beamline partners at the SLS for PX II and PX III
We invite companies and institutions to secure access to the beamlines X10SA/PX II and X06DA/PX III through a long term contract.