Grid-scale storage of electricity is vital in energy scenarios with a high share of renewable electricity generation, such as wind and solar power. Redox flow batteries are particularly suited for intra-day time-shifting storage applications, yet investment costs need to be lowered for economic viability of the technology. We demonstrate a new ion conducting membrane that improves shortcomings of currently used materials and is potentially cheaper to produce.
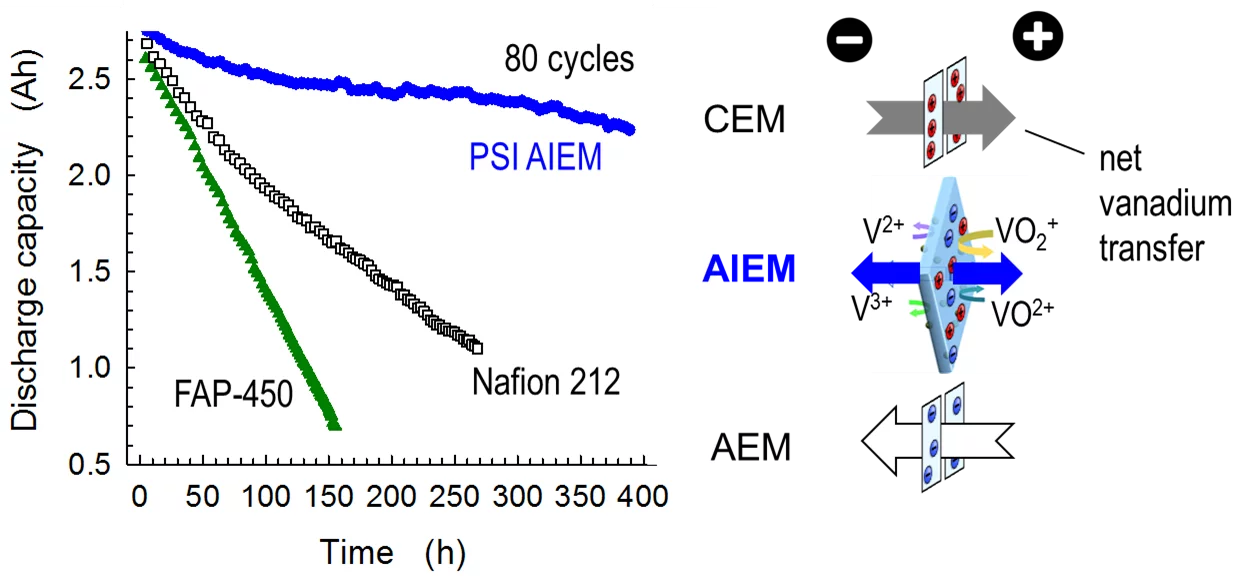
Vanadium redox flow batteries (VFB) use vanadium-ions in different oxidation states as redox-active species in the two electrolytes, thus irreversible contamination of electrolytes by cross-mixing is avoided. They offer independent scalability of energy and power, safe operation, long cycle-life and deep discharge capability. In the current state of the art, either a cation exchange membrane (CEM) or an anion exchange membrane (AEM) is used as a polymer electrolyte in VFBs. CEMs offer high conductivity, yet vanadium barrier properties are poor, and vice-versa for AEMs. Furthermore, with these membrane types, capacity fading is rather pronounced, which is related to the asymmetric transport properties of vanadium-ions through these materials. Therefore, amphoteric ion exchange membranes (AIEMs) were designed, which contain both anion and cation exchange groups, to balance transport effects. On the device level (cf. Figure), the superior capacity retention using this type of membrane is clearly demonstrated. Currently, the technology is further developed in the framework of a Bridge-Discovery project (http://p3.snf.ch/Project-176653) for scale-up and industrial implementation.
Contact
PD Dr. Lorenz GublerPaul Scherrer Institut
5232 Villligen PSI
Telephone: +41 56 310 26 73
E-mail: lorenz.gubler@psi.ch
Original Publication
Tackling capacity fading in vanadium flow batteries with amphoteric membranesOldenburg, F.J., Schmidt, T.J., and Gubler, L.,
J. Power Sources 368 (2017), 68-72
DOI: 10.1016/j.jpowsour.2017.09.051