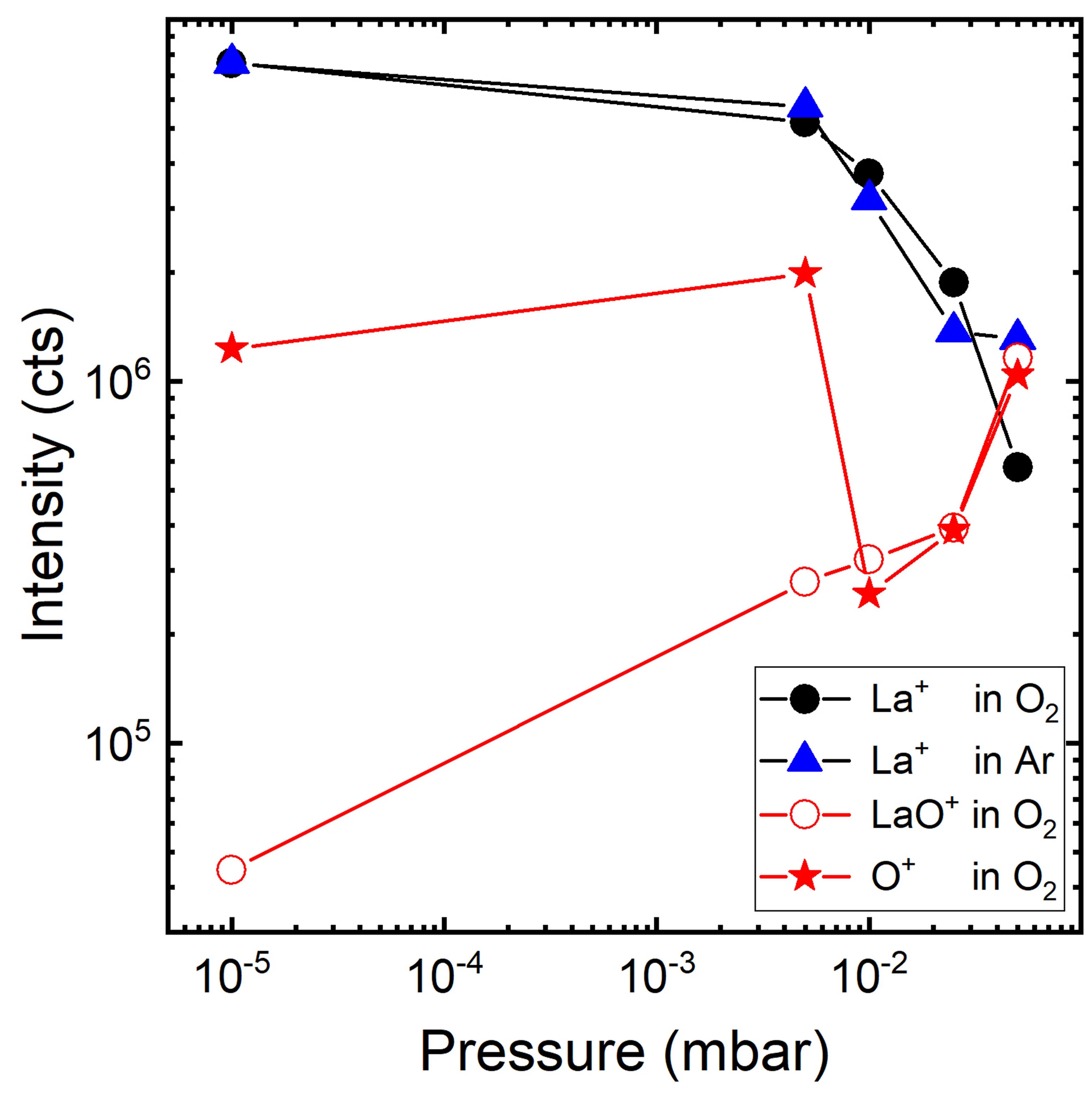
The gas-phase reaction dynamics and kinetics in a laser induced plasma are very much dependent on the interactions of the evaporated target material and the background gas. For metal (M) and metal–oxygen (MO) species ablated in an Ar and O2 background, the expansion dynamics in O2 are similar to the expansion dynamics in Ar for M+ ions with an MO+ dissociation energy smaller than O2. This is different for metal ions with an MO+ dissociation energy larger than for O2. This study shows that the plume expansion in O2 differentiates itself from the expansion in Ar due to the formation of MO+ species. It also shows that at a high oxygen background pressure, the preferred kinetic energy range to form MO species as a result of chemical reactions in an expanding plasma, is up to 5 eV.
Reference: Xiang et al., Materials 15, 4862 (2022)
Read full article: here