Electrophysiological methods and devices
for the activity measurement of membrane proteins
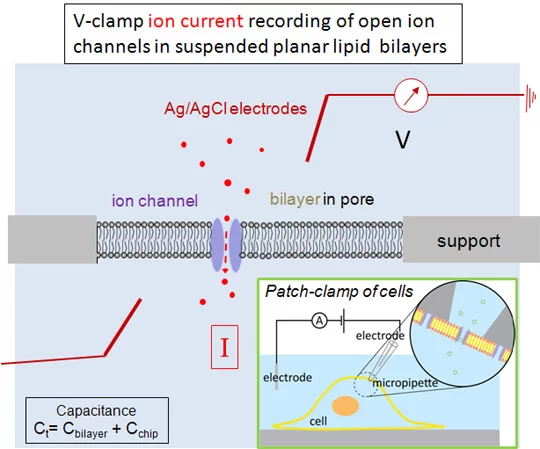
Translocation of ions and charged molecules across biological membranes are sensitively measured using electrophysiological instruments. In principle, an electrical potential is applied across a lipid bilayer membrane and, if ion channels are open or membrane transporters active, the resulting currents are measured. Membrane resistance in the Giga-Ohm range of lipid bilayers suspended in micro- or nanopores of the supporting material can be achieved, a prerequisite for sensitive measurements. Using impedance spectroscopy, the sealing resistance and the total capacitances of the bilayers and the supporting material are determined. As in patch-clamp measurements (see insert), voltage pulses can be applied to planar lipid bilayers and opening of ion channels determined as intermediate currents of some pico Ampères. To measure these low currents, measurements have to be carried out in a Faraday-cage which shields the measurement cell from outside disturbances.
For more information, contact Dr. Jinghui Luo
For more information, contact Dr. Jinghui Luo
Instrumentation
- Two instruments (Potentiostat Autolab, PG12) are available, equipped with an impedance and a low current module. Voltage-clamp measurements are carried out using an Axopatch 200 B instrument and a Digidata 1440 A analyser inside a Faraday cage (Warner) .
- Rotavap and extruders are used to generate liposomes of uniform sizes.
- Transport of solvents in microfluidic systems is achieved by computer-controlled NEMESYS syringe devices.
Materials
Fabrication, derivatization and analysis of micro- and nanopore materials are carried out in collaboration with PSI
Laboratory for micro- and nanotechnolgy and external partners.
Two kinds of materials have been used for bioanalytical devices: silicon and polymers. Silicon technology requires clean room laboratories including essentially photolithography or e-beam facilities for structure definition and etching processes for structure generation. Polymers can be structured by hot embossing and molding techniques and surfaces can be functionalized using chemical methods. Scanning electron microscopy, contact angle measurements and fluorescence microscopy are methods to analyse surface properties.
Laboratory for micro- and nanotechnolgy and external partners.
Two kinds of materials have been used for bioanalytical devices: silicon and polymers. Silicon technology requires clean room laboratories including essentially photolithography or e-beam facilities for structure definition and etching processes for structure generation. Polymers can be structured by hot embossing and molding techniques and surfaces can be functionalized using chemical methods. Scanning electron microscopy, contact angle measurements and fluorescence microscopy are methods to analyse surface properties.
Pore generation
In a 300 nm thin silicon nitride membrane on a silicon chip, pores of 200 nm to 25 micrometers have been made. For ion channel recordings one pore is sufficient, whereas for transporter protein recordings arrays of pores are required. Since the unitary translocation rate of transporters is about 1000 times lower than that of ion channels, a sufficient surface density of transporter molecules is required.
The quality of bilayer sealing depends on the lipid composition, the bilayer formation method, the material and the surface properties (roughness, contact angle). The capacitance mainly depends on the thickness of the supporting material. If it is thinner than about 10 micrometers, the capacitance values can be hardly compensated to the low values necessary for Voltage-clamp experiments.
The quality of bilayer sealing depends on the lipid composition, the bilayer formation method, the material and the surface properties (roughness, contact angle). The capacitance mainly depends on the thickness of the supporting material. If it is thinner than about 10 micrometers, the capacitance values can be hardly compensated to the low values necessary for Voltage-clamp experiments.
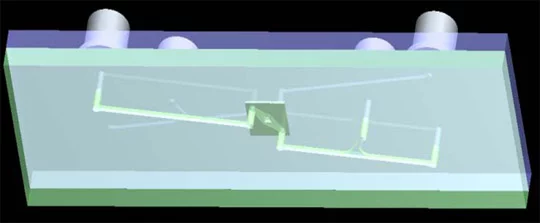
Microfluidic systems
Fluidic transport to the micrometer-sized pores is tricky. Solvents have to be transported, electrodes integrated and membrane protein activities of interest measured. Presently emphasis is put on making prototype devices of polymers.
Methods
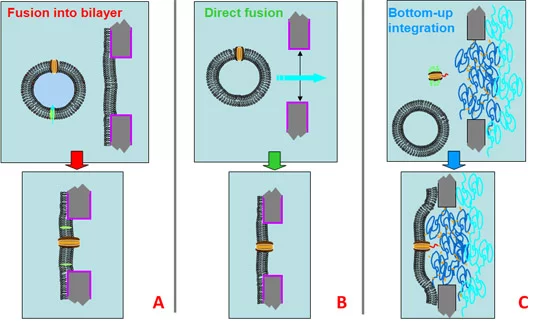
The integration of membrane proteins into lipid bilayer membranes suspended in micro- or nanopores is a critical step. Three different strategies (A, B or C) have been evaluated. Pore size, lipid composition and the bilayer formation method, influence the stability of the planar lipid proteobilayers. Electrophysiological methods are needed to monitor bilayer formation and to determine membrane-sealing resistance and capacitance of the bilayers formed.
Measuring activities: current traces
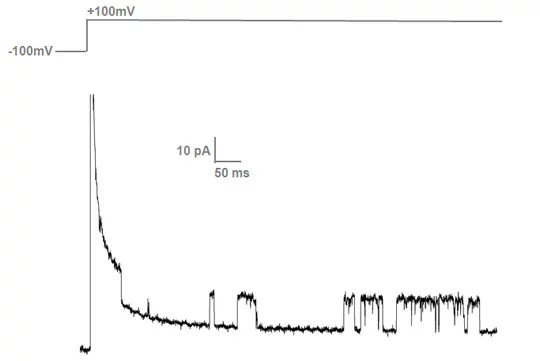
Single voltage-gated ion channels are activated by changing the potential from about - 150 mV to + 150 mV. Individual channels remain open for some ms and the opening and closing of channels can be recorded using the Axopatch instrument.
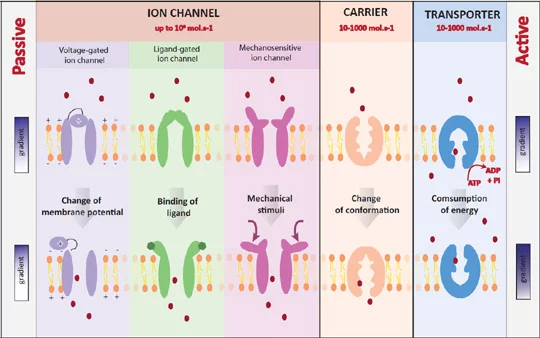
Membrane proteins of interest
Using electrophysiological methods, membrane protein mediated translocation of ions across bilayers can be monitored. Ion channels and transporters are important targets for drugs and thus highly relevant in drug discovery.