Scope of project
This laboratory-based research program is oriented towards fundamental research and is organized into a three-pronged scientific effort:
Kinetics of atmospheric trace gases on ices - The study of the chemical reactivity of atmospheric trace gases with pure and contaminated ice substrates under conditions where the solid substrate is controlled by FTIR, either in transmission or in grazing incidence. A roughly 1 cm2 ice substrate is condensed from the gas phase and doped under controlled conditions with HCl, HNO3 or organic molecules and subsequently probed by pure H2O vapor, HCl or HNO3 in order to measure the accommodation coefficients at temperatures of 150 to 220 K in a stirred flow reactor. Additional information may be obtained here for general information and here for details.
Surface characterization of complex interfaces (aerosols) using chemical titration with reactive gases - The surface characterization of complex organic or inorganic interfaces, such as soot, SOA, TiO2 or Ag nanoparticles, using reactive probe gases whose uptake is measured in a low pressure flow reactor (Knudsen cell). The idea behind is to "interrogate" surface functional groups of the exposed nanomaterial using reactive gases that specifically react with surface functional groups, for instance carboxylic acid groups on the surface are "titrated" with N(CH3)3 revealing the number of base molecules disappearing from the gas phase in the presence of the substrate. The vision of the project is that the particle composition reveals the origin, the surface composition using the present technique reveals the aging in terms of a change in the chemical reactivity of aerosol particles.
Elementary reactions of free radicals with halogen acids: need for revision of standard heats of formation of transient species important in combustion and atmospheric chemistry? - This project will investigate the chemical kinetics as a function of temperature of elementary reactions used to determine the thermodynamics of free radicals relevant to combustion and atmospheric chemistry. The type of free radical R such as CH3, t-C4H9, allyl (C3H5), benzyl (C7H7) will be reacted with a hydrohalic acid such as HBr or HI as follows: R + HX (X = Br, I) ==> R-H + X as a function of temperature in the range 300 to 700 K. When the thermochemical parameters (energy of activation Ea and preexponential factor A) are combined with the reverse reaction (existing literature values) the equilibrium constant may be calculated as a function of T. This will enable the extraction of the thermochemical parameters (standard enthalpy of formation and absolute entropy of R). The measurements are performed under molecular flow conditions using a Knudsen flow reactor coupled to either a VUV-single photon ionization or a molecular-beam modulated phase-sensitive electron impact quadrupole mass spectrometer.
Kinetics of atmospheric trace gases on ices - The study of the chemical reactivity of atmospheric trace gases with pure and contaminated ice substrates under conditions where the solid substrate is controlled by FTIR, either in transmission or in grazing incidence. A roughly 1 cm2 ice substrate is condensed from the gas phase and doped under controlled conditions with HCl, HNO3 or organic molecules and subsequently probed by pure H2O vapor, HCl or HNO3 in order to measure the accommodation coefficients at temperatures of 150 to 220 K in a stirred flow reactor. Additional information may be obtained here for general information and here for details.
Surface characterization of complex interfaces (aerosols) using chemical titration with reactive gases - The surface characterization of complex organic or inorganic interfaces, such as soot, SOA, TiO2 or Ag nanoparticles, using reactive probe gases whose uptake is measured in a low pressure flow reactor (Knudsen cell). The idea behind is to "interrogate" surface functional groups of the exposed nanomaterial using reactive gases that specifically react with surface functional groups, for instance carboxylic acid groups on the surface are "titrated" with N(CH3)3 revealing the number of base molecules disappearing from the gas phase in the presence of the substrate. The vision of the project is that the particle composition reveals the origin, the surface composition using the present technique reveals the aging in terms of a change in the chemical reactivity of aerosol particles.
Elementary reactions of free radicals with halogen acids: need for revision of standard heats of formation of transient species important in combustion and atmospheric chemistry? - This project will investigate the chemical kinetics as a function of temperature of elementary reactions used to determine the thermodynamics of free radicals relevant to combustion and atmospheric chemistry. The type of free radical R such as CH3, t-C4H9, allyl (C3H5), benzyl (C7H7) will be reacted with a hydrohalic acid such as HBr or HI as follows: R + HX (X = Br, I) ==> R-H + X as a function of temperature in the range 300 to 700 K. When the thermochemical parameters (energy of activation Ea and preexponential factor A) are combined with the reverse reaction (existing literature values) the equilibrium constant may be calculated as a function of T. This will enable the extraction of the thermochemical parameters (standard enthalpy of formation and absolute entropy of R). The measurements are performed under molecular flow conditions using a Knudsen flow reactor coupled to either a VUV-single photon ionization or a molecular-beam modulated phase-sensitive electron impact quadrupole mass spectrometer.
Key findings
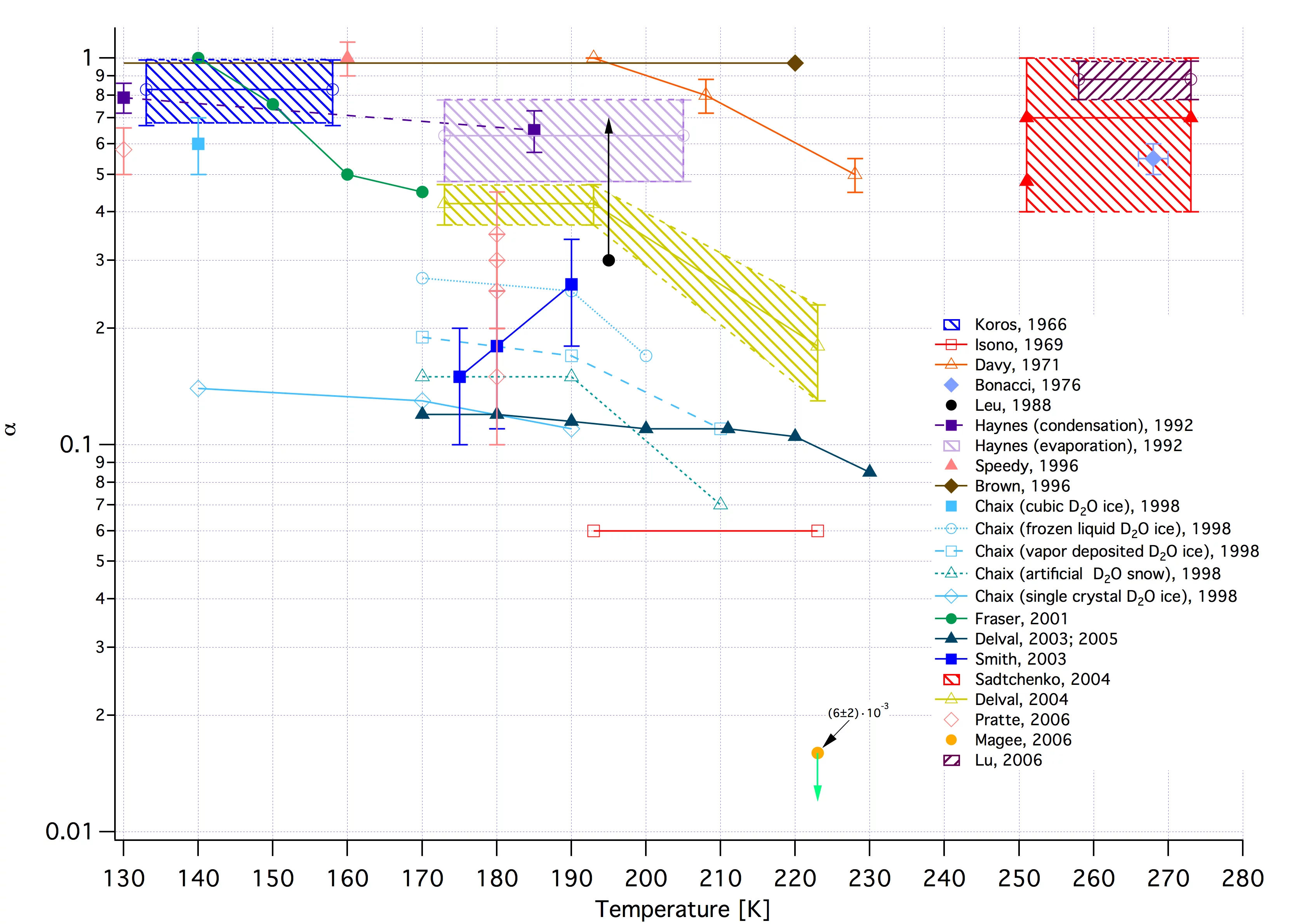
- At stratospherically relevant temperatures the T-dependence of the mass accommodation coefficient "alpha" for H2O vapor on pure and doped H2O-ice is smaller than the dependence of "alpha" on the type of ice (e.g. vapor-condensed, artifical snow or single crystal ice). See link to attached synopsis of results.
- The absolute magnitude of "alpha" for H2O vapor on H2O-ice depends on the degree of supersaturation, i.e on the type of experiment: "alpha" tends towards unity at high and levels off at approximately 0.2 at low H2O supersaturations.
- On soot there are many types of coexisting surface functional groups, including acidic and basic as well as strongly and weakly-reducing groups. This depends first and foremost on the combustion conditions, less so on the fuel used to generate the soot (flame soot in diffusion flame).
- A significant correlation of the types of surface functional groups with health markers of workers exposed to soot particulates in a bus maintenance yard with the number of strongly reducing surface groups was found. In addition, these reducing surface functional groups act as catalysts in the oxidation of reduced species (antioxidants) which amounts to approximately a 100 redox cycles with air oxygen. Similar results have been found for Ag nanoparticles that present Ag+ sites on their surface and also enable catalytic redox cycling.
Publications
Representative (peer-reviewed) Journal Articles
Probing functional groups at the gas-aerosol interface using heterogeneous titration reactions: A tool for predicting aerosol health effects? .A. Setyan, J.-J. Sauvain, M. Guillemin, M. Riediker, B. Demirdjian and M. J. Rossi, CHEMPHYSCHEM 2010, 11, 3823-3835;
Article
The influence of monolayer amounts of HNO3 on the evaporation rate of H2O over ice at 179 ≤ T/K ≤ 208: A quartz crystal microbalance study.
Ch. Delval and M. J. Rossi, J. Phys. Chem. A 2005, 109, 7151-7165.
Article
An Overview of current issues in the uptake of atmospheric trace gases by aerosols and clouds.
C.E. Kolb, R.A. Cox (eds.), J.P.D. Abbatt, M. Ammann, E.J. Davis, D.J. Donaldson, B.C. Garrett, C. George, P. Griffiths, D.F. Hanson, M. Kulmala, G. McFiggans, U. Pöschl, I. Riipinen, M.J. Rossi, Y. Rudich, P.E. Wagner, P.M. Winkler, D.R. Worsnop and C.D. O’Dowd, Atmos. Chem. Phys. Discuss. 2010, 10, 11139-11250; Atmos. Chem. Phys. 2010, 10, 10561-10605.
Article