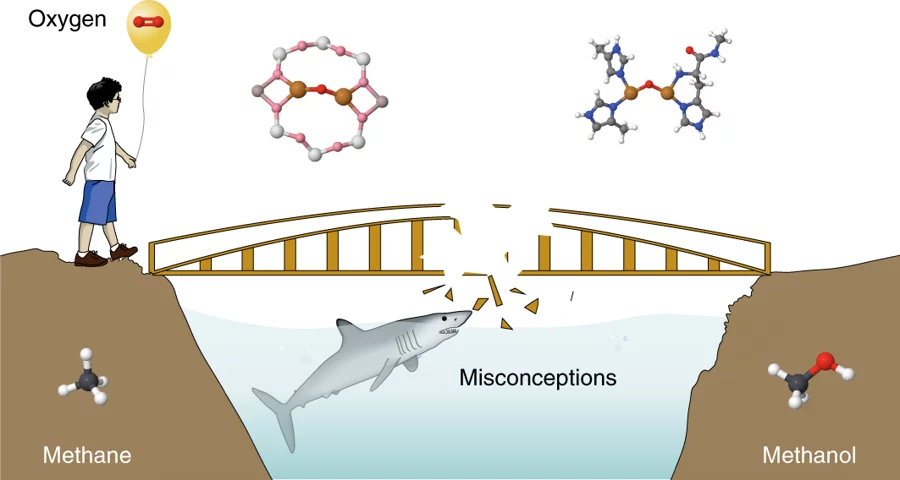
It needs no introduction that crude oil reserves are fundamental to the functioning of our automobiles and public transportation systems. However, at the sites that generate the fuel for our vehicles, associated natural gas is merely flared to carbon dioxide and released to the atmosphere. The volume of such gas flares worldwide amounts to more than a hundred billion cubic meters, a corresponding market value of $20 billion, and one might wonder how injudicious we have been in handling a resource. Nevertheless, the flaring of natural gas is more a reflection of the difficulty in its valorization rather than a lack of desire to do so. The long-standing ambition has been to figure out a more prudent use of methane. The on-site conversion of methane - the principal component of natural gas - to methanol is an attractive and, at the same time, challenging route. Interestingly, bacteria can perform the transformation of methane to methanol in a highly efficient way. So can we design a process that truly can mimic the performance of bacteria and enzymes? Can a direct methane-to-methanol process be industrially feasible in the near future? Which are the most promising approaches and what are the kind of systems that need to be engineered? Answers to such questions and more form the basis of this perspective article in Nature Catalysis.
Additional Information
Contact
Prof. Dr. Jeroen A. van Bokhoven
Laboratory Leader
Paul Scherrer Institut
Telephone: +41 56 310 5046
E-mail: jeroen.vanbokhoven@psi.ch
and
Professor for Heterogeneous Catalysis
ETH Zurich
Telephone: +41 44 632 5542
E-mail: jeroen.vanbokhoven@chem.ethz.ch
Original Publication
Misconceptions and challenges in methane-to-methanol over transition-metal-exchanged zeolites
DOI: 10.1038/s41929-019-0273-z