X-ray photoelectron spectroscopy (XPS), also known as electron spectroscopy for chemical analysis (ESCA), is a prime tool to study the local chemical environment of molecules or materials at the atomic level. Kai Siegbahn was awarded the Nobel Prize in Physics in 1981 for his pioneering contribution and understanding of chemical shifts in XPS experiments. Capitalizing on the atomic selectivity of X-rays, XPS spectroscopy has become one of the widely used techniques for chemical analysis.
A dream application for XPS is to expand the approach from characterizing static samples to following chemical reactions and inner atomic processes as they are taking place. The advent of X-ray free-electron lasers (XFELs) has opened the possibility to study processes with X-rays down to the femtosecond time domain and thus explore out of the equilibrium dynamics with temporal resolution of just a few femtoseconds (10-15 s). With such a resolution, nuclear rearrangements and even the preceding electron dynamics during a chemical reaction could in principle be probed. Now, one can envision to observe chemical changes right after light excitation, which opens a new avenue for understanding of light-harvesting complexes, radiation damage, and photochemical processes.
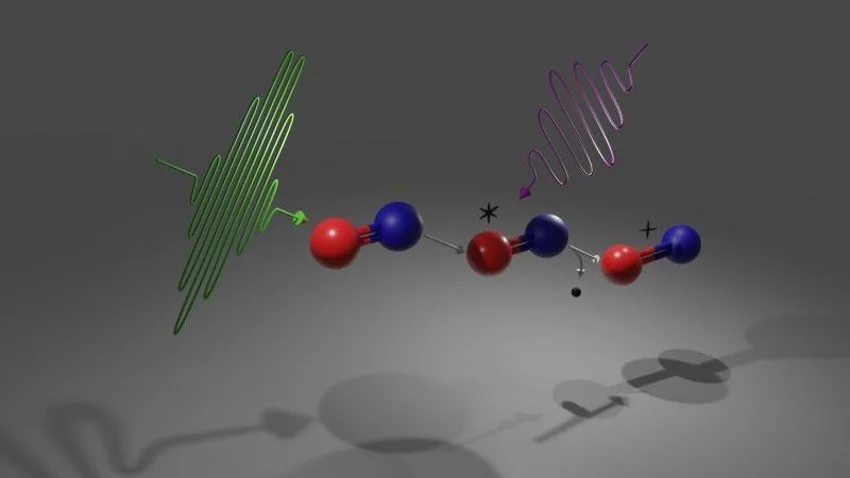
In the work “Observation of site-selective chemical bond changes via ultrafast chemical shifts”, an international group of researchers expanded time resolved XPS in two aspects. They employed an ultrashort x-ray pulse to start a reaction at a specific atomic site and subsequently followed the reaction at a different site of a molecule. With this so-called x-ray pump, x-ray probe approached they tracked the electronic and nuclear dynamics in prototypical carbon monoxide (CO) molecules using time resolved XPS. More specifically, the first x-ray pulse was tuned to exclusive excited the core-electrons of oxygen in the CO molecule, while the second x-ray pulse probed the chemical environment sensed by the carbon atom in the following few femtoseconds. The experiment was so sensitive that even the first electronic excitation of the oxygen atom and subsequent Auger decay, one of the fastest atomic processes, was observed in the experiments. In the following, the chemical bond between the atoms broke and the molecule fragmented. The chemical shifts of the carbon innershell electrons could reflected the complex dynamics of the molecule with femtoseconds precision. The reported experimental and theoretical advancements pave the way for future experiments at XFELs and move closer towards the dream of resolving local chemical bond changes in real time. Future experiments will allow to follow ultrafast energy and charge transport processes in complex molecular systems in real time, helping to improve a wide range of applications from solar harvesting materials to light triggered pharmeceuticals.
The work was performed by an international collaborations with groups from EPFL and PSI (Switzerland), UAM (Madrid), ANL (USA), and Stanford (USA).
Andre Al-Haddad, Solène Oberli, Jesús González-Vázquez, Maximilian Bucher, Gilles Doumy, Phay Ho, Jacek Krzywinski, Thomas J. Lane, Alberto Lutman, Agostino Marinelli, Timothy J. Maxwell, Stefan Moeller, Stephen T. Pratt, Dipanwita Ray, Ron Shepard, Stephen H. Southworth, Alvaro Vazquez-Mayagoitia, Peter Walter, Linda Young, Antonio Picón, and Christoph Bostedt, “Observation of site-selective chemical bond changes via ultrafast chemical shifts”, Nature Communications 13, 7170 (2022)