Lab News & Scientific Highlights
Chemical Imaging to Spy on Malaria Parasites
Unique insights into the adolescence and metabolism of a Malaria parasite in a human red blood cell are obtained by a new chemical imaging methodology – in situ correlative X-ray fluorescence microscopy and soft X-ray tomography.
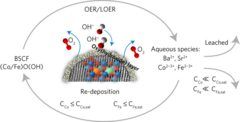
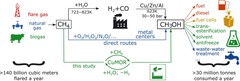
Nanomaterial helps store solar energy: efficiently and inexpensively
By combining a scalable cutting-edge synthesis method with time-resolved X-ray absorption spectroscopy measurements, it was possible to capture the dynamic local electronic and geometric structure during realistic operando conditions for highly active OER perovskite nanocatalysts.
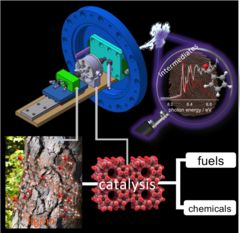
Understanding the reaction mechanism in lignin catalytic fast pyrolysis
Lignin is a major constituent of plants, and may be used as a precursor for fuels and fine chemicals. Catalytic fast pyrolysis of lignin is one of the most promising approaches. By using vacuum ultraviolet synchrotron radiation and threshold photoelectron spectroscopy we could identify elusive intermediates, which are responsible for the formation of phenol and benzene and could thus tackle this reaction mechanism. Mechanistic understanding could enable targeted improvement of production methods in the future, beyond the currently used "cook-and-look" approach.
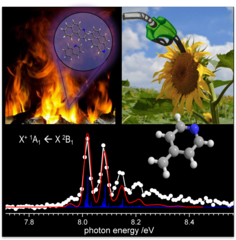
Isomer-Selective Generation and Spectroscopic Characterization of Biofuel Intermediates
Online combustion analysis relies heavily on spectral data to detect reactive intermediates isomer-selectively to establish e.g. kinetic flame models. Due to the difficulty to generate these species cleanly, spectral data are rather scarce. Here we report on the selective generation of three picolyl radical isomers (C5H4N-CH2*) by deamination of aminomethylpyridines. Picolyl radicals are relevant in biofuel combustion, and could now be characterized by threshold photoelectron spectroscopy using synchrotron radiation. Vibrationally resolved bands and distinct ionization energies allow for isomer-specific detection of these elusive species in complex environments and permit us to explore new avenues in soot- and NOx formation kinetics.
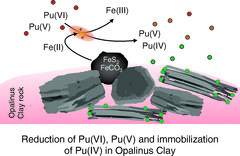
Fate of Plutonium through a Geological Reactive Barrier
Natural geological and engineered barriers play a key role in protecting the environment and the anthroposphere from the hazardous impact of deposited waste or spreading contaminants. Such natural geological and engineered barrier materials are commonly complex and heterogeneous. In-situ multimodal microscopic studies under conditions relevant to deep geological formations are crucial to identify the reactive components and reaction pathways or to validate proposed immobilization mechanisms. The present study demonstrated that a simplistic description by a sole reactive component is not an adequate representation of the geochemical reactivity responsible for the immobilization of plutonium within a natural Clay Rock barrier. Multimodal chemical imaging studies on intact, undisturbed systems are absolutely essential to ascertain the geochemical reactivity for relevant geochemical conditions and settings.
Selective anaerobic oxidation of methane enables direct synthesis of methanol
On the basis of in situ x-ray absorption spectroscopy, infrared spectroscopy, and density functional theory calculations, it was proposed a mechanism involving methane oxidation at Cu II oxide active centers, followed by Cu I reoxidation by water with concurrent formation of hydrogen.
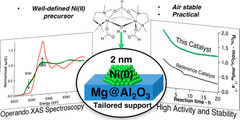
Molecularly Tailored Nickel Precursor and Support Yield a Stable Methane Dry Reforming Catalyst with Superior Metal Utilization
The superior performance of molecularly tailored methane dry reforming catalyst resulted in a maximization of the amount of accessible metallic nickel in the form of small nanoparticles preventing coke deposition. Operando X-ray absorption near-edge structure spectroscopy confirms that deactivation largely occurs through the migration of Ni into the support.
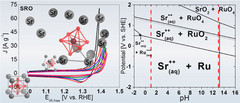
Unraveling Thermodynamics, Stability, and Oxygen Evolution Activity of Strontium Ruthenium Perovskite Oxide
Ru-based perovskites, i.e. SrRuO3 and LaRuO3, have been predicted as active perovskites to exhibit a particularly high oxygen evolution reaction activity. We highlight that understanding the origin of stability under a real operating environment is absolutely essential for the design of a sustainable electrocatalyst with optimal balance between activity and stability.
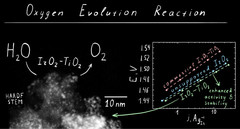
IrO2‑TiO2: A High-Surface-Area, Active, and Stable Electrocatalyst for the Oxygen Evolution Reaction
We have developed a synthetic approach to highsurface-area chlorine-free iridium oxide nanoparticles dispersed in titania (IrO2-TiO2), which is a highly active and stable OER catalyst in acidic media. Operando X-ray absorption studies demonstrate the evolution of the surface species as a function of the applied potential, suggesting the conversion of the initial hydroxo surface layer to the oxo-terminated surface via anodic oxidation.