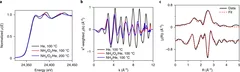
The selective catalytic oxidation of NH3 to N2 presents a promising solution for the abatement of unused NH3-based reductants from diesel exhaust after treatment. Supported Pd nanoparticle catalysts show selectivity to N2 rather than NOx, which is investigated in this work. The link between Pd nanoparticle structure and surface reactivity was found using operando X-ray absorption fine structure spectroscopy, diffuse reflectance infrared Fourier-transformed spectroscopy and on-line mass spectrometry. Nitrogen insertion into the metallic Pd nanoparticle structure at low temperatures (<200 °C) was found to be responsible for high N2 selectivity, whereas the unfavourable formation of NO is linked to adsorbed nitrates, which form at the surface of bulk PdO nanoparticles at high temperatures (>280 °C). Our work demonstrates the ability of combined operando spectroscopy and density functional theory calculations to characterize a previously unidentified PdNx species, and clarify the selectivity-directing structure of supported Pd catalysts for the selective catalytic oxidation of NH3 to N2.
Contact
Dr Maarten Nachtegaal
SuperXAS beamline
Laboratory for Synchrotron Radiation and Femtochemistry (LSF)
Swiss Light Source, Paul Scherrer Intitute
5232 Villigen-PSI, Switzerland
Telephone: +41 56 310 30 56
E-mail: marten.nachtegaal@psi.ch
Original Publication
Structural selectivity of supported Pd nanoparticles for catalytic NH3 oxidation resolved using combined operando spectroscopy
Ellie K. Dann, Emma K. Gibson, Rachel H. Blackmore, C. Richard A. Catlow, Paul Collier, Arunabhiram Chutia, Tugce Eralp Erden, Christopher Hardacre, Anna Kroner, Maarten Nachtegaal, Agnes Raj, Scott M. Rogers, S. F. Rebecca Taylor, Paul Thompson, George F. Tierney, Constantinos D. Zeinalipour-Yazdi, Alexandre Goguet & Peter P. Wells
Nature Catalysis, 28 January 2019
DOI: 10.1038/s41929-018-0213-3