Operando X-ray Spectroscopy
The Operando X-ray Spectroscopy belongs to Environment Division (ENE) of PSI and is within a matrix organization responsible for the operation of the SuperXAS and Debye beamlines to LSF.
Scientific Highlights
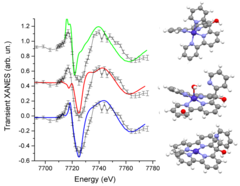
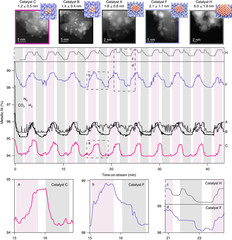
Structure of the Co(I) Intermediate of a Cobalt Pentapyridyl Catalyst for Hydrogen Evolution Revealed by Time‐Resolved X‐ray Spectroscopy
The mechanism of hydrogen evolution by cobalt polypyridyls catalysts is investigated. Pump-probe X‐ray absorption spectra measured at SuperXAS in the microsecond time range indicate that the pendant pyridine dissociates from the cobalt in the intermediate Co(I) state. This opens the possibility for pyridinium to act as an intramolecular proton donor, which can be used for the development of efficient catalysts.
Stable complete methane oxidation over palladium based zeolite catalysts
Using targeted synthesis and in situ characterization a palladium catalyst with improved stability against sintering during methane oxidation was prepared.
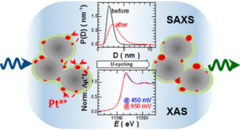
Combining SAXS and XAS To Study the Operando Degradation of Carbon-Supported Pt-Nanoparticle Fuel Cell Catalysts
In the last two decades, small-angle X-ray scattering (SAXS) and X-ray absorption spectroscopy (XAS) have evolved into two well-established techniques capable of providing complementary and operando information about a sample’s morphology and composition, respectively. Considering that operation conditions can often lead to simultaneous and related changes in a catalyst’s speciation and shape, herein we introduce a setup that combines SAXS and XAS in a configuration that allows optimum acquisition and corresponding data quality for both techniques.
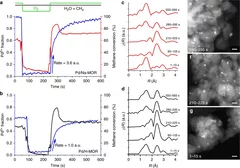
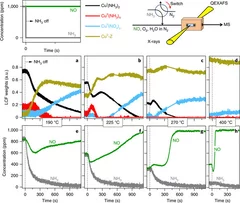
Time-resolved copper speciation during selective catalytic reduction of NO on Cu-SSZ-13
Through the combination of time-resolved X-ray absorption spectroscopy and transient experimentation, we were able to capture an ammonia inhibition effect on the rate-limiting copper re-oxidation at low temperature.
Unravelling structure sensitivity in CO2 hydrogenation over nickel
Using a unique set of well-defined silica-supported Ni nanoclusters (1–7 nm) and advanced characterization methods it was proved how structure sensitivity influences the mechanism of catalytic CO2 reduction, the nature of which has been long debated.
Nanomaterial helps store solar energy: efficiently and inexpensively
By combining a scalable cutting-edge synthesis method with time-resolved X-ray absorption spectroscopy measurements, it was possible to capture the dynamic local electronic and geometric structure during realistic operando conditions for highly active OER perovskite nanocatalysts.
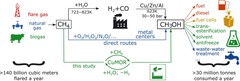
Selective anaerobic oxidation of methane enables direct synthesis of methanol
On the basis of in situ x-ray absorption spectroscopy, infrared spectroscopy, and density functional theory calculations, it was proposed a mechanism involving methane oxidation at Cu II oxide active centers, followed by Cu I reoxidation by water with concurrent formation of hydrogen.
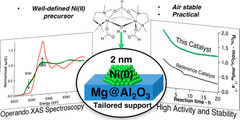
Molecularly Tailored Nickel Precursor and Support Yield a Stable Methane Dry Reforming Catalyst with Superior Metal Utilization
The superior performance of molecularly tailored methane dry reforming catalyst resulted in a maximization of the amount of accessible metallic nickel in the form of small nanoparticles preventing coke deposition. Operando X-ray absorption near-edge structure spectroscopy confirms that deactivation largely occurs through the migration of Ni into the support.
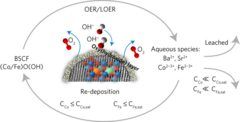
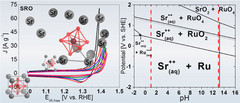
Unraveling Thermodynamics, Stability, and Oxygen Evolution Activity of Strontium Ruthenium Perovskite Oxide
Ru-based perovskites, i.e. SrRuO3 and LaRuO3, have been predicted as active perovskites to exhibit a particularly high oxygen evolution reaction activity. We highlight that understanding the origin of stability under a real operating environment is absolutely essential for the design of a sustainable electrocatalyst with optimal balance between activity and stability.