Welcome to the Electrochemistry Laboratory (LEC)
The Electrochemistry Laboratory (LEC), established 1988, is part of the Energy and Environment Research Division (ENE) at the Paul Scherrer Institute. The laboratory comprises 5 interacting research groups that deal with almost all aspects of electrochemical energy storage and conversion. PSI’s Electrochemistry Laboratory is Switzerland’s largest Center for Electrochemical Research.
Lab News & Scientific Highlights
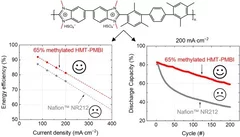
Polybenzimidazole Membrane Design Principles for Vanadium Redox Flow Batteries
Energy storage technologies with long storage duration are essential to stabilize electricity grids with a high share of intermittent renewable power. In a redox flow battery, the electrochemical conversion unit, where the charging and discharging reaction takes place, is spatially separated from the energy storage medium. In the all-vanadium redox flow battery (VRFB), a sulfuric acid aqueous electrolyte with dissolved vanadium ions is used as the storage medium. Vanadium is present in 4 different oxidation states, the redox couple vanadium(II) and (III) on the negative side of the cell, and vanadium(IV) and (V) on the positive side. This allows the battery to be repeatedly charged and discharged. A separator or membrane is used between the negative and positive electrode, which should selectively conduct the ions of the supporting electrolyte and minimize the passage of vanadium ions. Fluorinated membranes, such as Nafion™, are often used for this key component, but these ionomers were not originally developed for this application and therefore have functional shortcomings. Furthermore, the production and use of fluorinated materials is to be severely restricted or even banned in Europe. Therefore, the development of hydrocarbon-based membranes for the VRFB is of great importance. The study reported here focuses on polybenzimidazole polymers and membranes, which could be a promising materials class for next generation flow batteries.
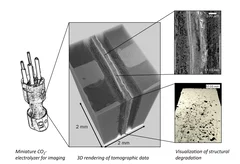
Unraveling degradation processes in a bipolar membrane CO2 electrolyzer by time-resolved X-ray tomographic microscopy
Employing a bipolar ion conducting membrane (BPM) in forward bias is a convenient solution for the biggest issues in the more common anion exchange membrane (AEM) CO2 co-electrolysis: the degradation of the performance caused by carbonate salt precipitation at the cathode and the decrease of net CO2 conversion caused by the crossover of this molecule from cathode to anode also requiring energy for downstream gas separation. However, the performance and stability of this device remain largely insufficient when using such a BPM configuration. To understand the reasons for this, we performed time-resolved X-ray tomographic microscopy of an operating BPM CO2 electrolyzer. The imaging method reveals partly unexpected degradation processes that result in design recommendations for improvement.
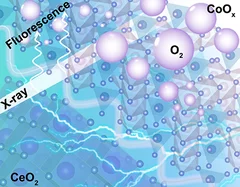
Insights into the superior oxygen evolution reaction activity of CoOx/CeO2 composite electrocatalyst
CeO2 significantly enhances the oxygen evolution reaction (OER) activity of CoOx, although the mechanism behind this synergy is still unclear. Here, operando hard X-ray absorption spectroscopy (hXAS) is applied to monitor the Co-K edge and Ce L3 edge in CoOx/CeO2 to shed light on the evolution of Co and Ce oxidation states during OER. In addition, ex situ soft XAS (sXAS) characterizations provide information on the irreversible surface-specific transformations of the Co L3 edge as well as the O K edge.
Upcoming Events
LEC on TV and Radio
Paul Scherrer Institut entwickelt Akku der Zukunft
Swiss Radio: SRF Regionaljournal Aargau Solothurn, May 8, 2017, 17:30h
Knopfbatterien im Test: Enorme Preisunterschiede
Swiss TV: Kassensturz, January 17, 2017, 21:10h
Die Renaissance der Batterie
Swiss TV: Einstein, November 26, 2015, 21:07h
Mobilität ohne Treibhausgase
Swiss TV: Einstein, November 21, 2013, 21:07h
Wasserstofftankstellen für die Schweiz?
Swiss TV: Einstein, November 21, 2013, 21:07h