The SINQ neutron source
Producing neutrons – the spallation reaction
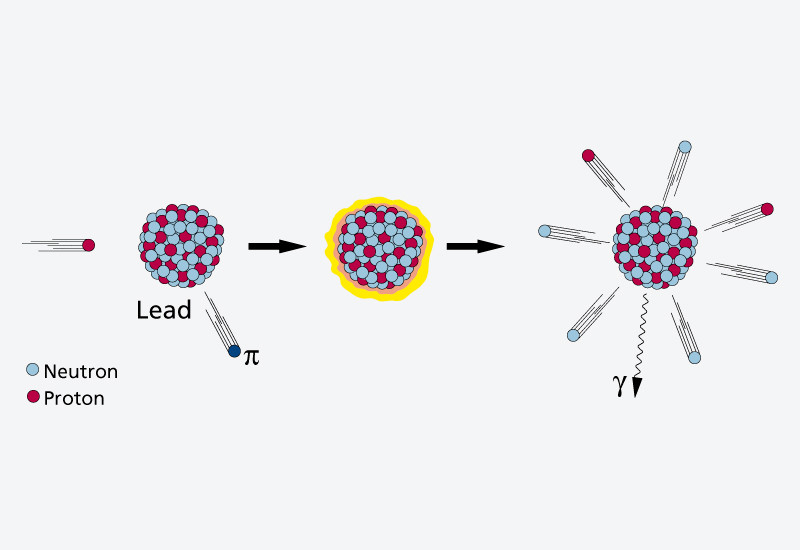
In the SINQ spallation source a beam of fast protons (at about 80 % of the speed of light) from the PSI proton accelerator facility strikes a block of lead (the Target). If a fast proton collides with a lead nucleus, the nucleus will be heated up and eject 10 to 20 neutrons.
To be used in experiments, neutrons have to be decelerated
The neutrons set free by this method are extremely fast – much too fast for the experiments. In order to decelerate the neutrons after they have been generated, the whole target is placed in a tank filled with heavy water(1). The neutrons are decelerated by collisions with the nuclei of the heavy hydrogen in this water. This produces thermal neutrons, which can be used to determine the structure of crystals, amongst other things.
Cold neutrons – investigations of larger structures require particularly slow neutrons
If larger structures – such as nanoparticles – are to be investigated using neutrons, these neutrons will have to be even slower (known as cold
neutrons).
This is because neutrons can be more successfully used in experiments if their wavelengths are approximately the same size as the structure being examined, and slow neutrons have a larger wavelength than fast ones.
An additional tank, with extremely cold heavy hydrogen (minus 250 degrees Celsius), has been integrated into the SINQ water tank in order to decelerate the neutrons even further.
Neutron guides channel the neutrons to the experiments
The neutron scattering instruments radiate from the neutron source. The neutrons travel to the various measuring stations via neutron guides
, which are glass conduits – coated internally with a special material – in which the neutrons are reflected repeatedly and thus transported to the experiment.
Notes
1: Heavy water – heavy hydrogen: The atomic nucleus of normal hydrogen consists of only one proton, while that of heavy hydrogen (deuterium) comprises one proton and one neutron. Molecules of heavy water contain heavy hydrogen, and heavy hydrogen is particularly suitable for neutron deceleration because it hardly absorbs any of the neutrons– unlike normal
hydrogen.