Scientific Highlights
TEY-STXM confirms homogeneous doping of nanoparticles for non-fullerene organic solar cells
One of the challenges in modern research on the fabrication of non-fullerene acceptor based organic solar cells is the availability of very efficient hole transport layers (HTLs). A new approach that avoids mutual solubility issues is to deposit the HTL from a suspension of doped organic nanoparticles. Surface-sensitive TEY-STXM measurements at the PolLux beamline characterised the homogeneity of the dopant in the nanoparticles and develop efficient nanoparticle HTL materials for organic solar cells.
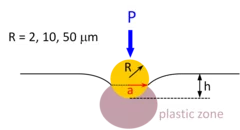
Investigations of the irradiation hardening on a ferritic model alloy from spherical nano-indentations
The objective of this project was to determine the contribution from a variety of obstacles to moving dislocations to the nano-indentation stress necessary to initiate plastic flow. The obstacles are characterized by different length scales. Among these characteristic lengths, there are those associated with the material microstructure such as grain size, dislocations density, irradiation-induced defects, and those related to the size of the plastic zone beneath the indenter, or equivalently to the size of the indent. Thus, we can classify the size effects into two categories: structural size effect and indentation size effect (ISE). The underlying idea is to quantify and separate these two effects on the unirradiated material first to be able to properly isolate the contribution of the irradiation defect on the measured hardness from the tests on irradiated materials.
Using quantum computers already today
Analogue quantum computers make ultrafast chemical reactions observable.
Open Quantum Institute launch
Dr. Cornelius Hempel, head of the Ion Trap Quantum Computation group at LNQ’s ETHZ-PSI Quantum Computing Hub, spoke to SRF to explain how quantum computers work and how future versions of these devices can be used to solve some of the big problems of our time.
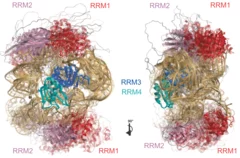
Integrative solution structure of PTBP1-IRES complex reveals strong compaction and ordering with residual conformational flexibility
RNA-binding proteins (RBPs) are crucial regulators of gene expression, often composed of defined domains interspersed with flexible, intrinsically disordered regions. Determining the structure of ribonucleoprotein (RNP) complexes involving such RBPs necessitates integrative structural modeling due to their lack of a single stable state. In this study, we integrate magnetic resonance, mass spectrometry, and small-angle scattering data to determine the solution structure of ...
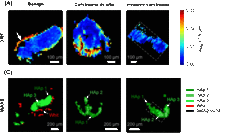
Whitlockite in mammary microcalcifications is not associated with breast cancer
Microcalcifications, small deposits of calcium-containing minerals that form in breast tissue, are often, but not always, a warning sign of breast cancer. The relationship between microcalcifications and cancer has not been fully understood thus far. Researchers discovered now that the relationship between microcalcifications and tumors seems to be linked to the presence of a particular mineral called whitlockite, which is rich in magnesium and is found in microcalcifications only in the absence of tumors.
LXN post-doctoral researcher Dr. Prajith Karadan wins Best Poster Award at MNE conference 2023, Berlin
The MNE (Micro and Nano Engineering) conference is a prestigious annual event that serves as a global platform for experts, researchers, and innovators in the field of micro and nanotechnology. This conference brings together leading minds from academia and industry to share cutting-edge research, exchange ideas, and explore emerging trends and breakthroughs in the world of micro and nanoengineering.
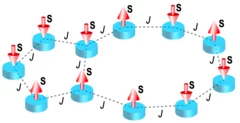
Electrically programmable magnetic coupling in an Ising network exploiting solid-state ionic gating
Two-dimensional arrays of magnetically coupled nanomagnets provide a mesoscopic platform for exploring collective phenomena as well as realizing a broad range of spintronic devices. In particular, the magnetic coupling plays a critical role in determining the nature of the cooperative behavior and providing new functionalities in nanomagnet-based devices. Here, we create coupled Ising-like nanomagnets ...
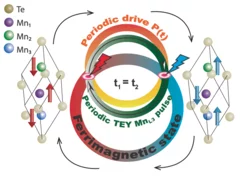
Efficient magnetic switching in a correlated spin glass
The interplay between spin-orbit interaction and magnetic order is one of the most active research fields in condensed matter physics and drives the search for materials with novel, and tunable, magnetic and spin properties. Here we report on a variety of unique and unexpected observations in thin multiferroic Ge1−xMnxTe films.
Insights into radical induced degradation of anion exchange membrane constituents
Electrochemical energy conversion devices, such as fuel cells and electrolyzers, using an anion exchange membrane (AEM) operating in the alkaline regime offer the prospect of the use of non-noble metal electrocatalysts and lower-cost cell construction materials. The wide-spread application of electrochemical cells with AEMs has been largely limited by the low chemical stability of the material. AEM degradation is triggered by i) nucleophilic attack by OH−, and ii) by reaction with free radicals formed during cell operation. Whereas the alkaline stability of AEMs has been greatly increased over the last 10 years, the understanding of mechanisms of radical induced degradation is limited. In this study, we have addressed this topic for the first time.