Scientific Highlights
Making a valuable resource usable with water
In oil extraction sites, gaseous methane is simply burned, even though it could actually be a useful precursor material for fuels and products of the chemical industry. One way to make methane usable is to convert it to methanol. Researchers at the Paul Scherrer Institute PSI and ETH Zurich have now developed a new chemical process that allows this conversion in an efficient and inexpensive way.
Cover page of CHEMCATCHEM
A paper titled "Optimization of the Reaction Conditions for Catalytic Fast Pyrolysis of Pretreated Lignin over Zeolite for the Production of Phenol" by Zhiqiang Ma is published in ChemCatChem and made it on the cover of issue 6/2017.
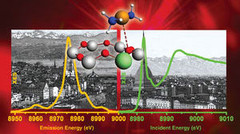
Nanotechnology enables new insights into chemical reactions
Eighty percent of all products of the chemical industry are manufactured with catalytic processes. Catalysis is also indispensable in energy conversion and treatment of exhaust gases. Industry is always testing new substances and arrangements that could lead to new and better catalytic processes. Researchers of the Paul Scherrer Institute PSI in Villigen and ETH Zurich have now developed a method for improving the precision of such experiments, which may speed up the search for optimal solutions.
Using methane rather than flaring it
Chemists at ETH Zurich and the Paul Scherrer Institute have found a new, direct way to convert gaseous methane into liquid methanol. This offers industry the interesting prospect of using the gas, rather than simply burning it off, as is currently the case.
Textbook on XAS and XES
During the last two decades, remarkable and often spectacular progress has been made in the methodological and instrumental aspects of x–ray absorption and emission spectroscopy. This progress includes considerable technological improvements in the design and production of detectors especially with the development and expansion of large-scale synchrotron reactors All this has resulted in improved analytical performance and new applications, as well as in the perspective of a dramatic enhancement in the potential of x–ray based analysis techniques for the near future.
Controlling tunnelling in methane loss from acetone ions by deuteration
If a ball is rolled up a hill with less kinetic energy than the potential energy at the top, it will return eventually, and stays bound in the valley. Tunnelling is a distinctly quantum mechanical phenomenon, in which such balls can magically cross the hill, and appear in the neighbouring valley, as if going through a tunnel. In order for this to happen with a non-negligible probability, the ball has to be small and the barrier, i.e. the hill, sharp.
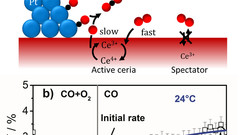
Catalytically Active and Spectator Ce3+ in Ceria-Supported Metal Catalysts
Identification of active species and the rate-determining reaction steps are crucial for optimizing the performance of oxygen-storage materials, which play an important role in catalysts lowering automotive emissions, as electrode materials for fuel cells, and as antioxidants in biomedicine. We demonstrated that active Ce3+ species in a ceria-supported platinum catalyst during CO oxidation are short-lived and therefore cannot be observed under steady-state conditions.
Metal organic frameworks for photo-catalytic water splitting
Growing experimental and computational evidence suggests that metal organic frameworks (MOFs) can make a meaningful contribution to catalytically promoted water splitting. They offer an impressive physical, spatial, chemical and electronic mutability with which to support and sustain water splitting half reactions. Their classical features à thermal stability, large surface area, high porosity and modularity à define them as versatile solid supports.
On the protonation of water
Imaging photoelectron photoion coincidence (iPEPICO) spectroscopy on isolated water molecules and water dimers establishes a new route to determining the water proton affinity (PA) with unprecedented accuracy. A floating thermochemical cycle constructed from the OH+ and H3O+ appearance energies and three other spectroscopic values establishes the water PA as 683.22 ± 0.25 kJ mol−1 at 0 K, which converts to 688.81 ± 0.25 kJ mol−1 at room temperature.