Scientific Highlights
Ultrafast structural changes direct the first molecular events of vision
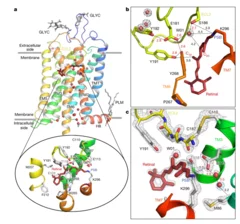
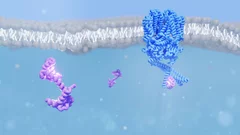
The visual pigment rhodopsin plays a critical role in the process of low-light vision in vertebrates. It is present in the disk membranes of rod cells in the retina and is responsible for transforming the absorption of light into a physiological signal. Rhodopsin has a unique structure that consists of seven transmembrane (TM) α-helices with an 11-cis retinal chromophore covalently bound to the Lysine sidechain of 7th TM helix. A negatively charged amino acid (glutamate) forms a salt bridge with the protonated Schiff base (PSB) of the chromophore to stabilize the receptor in the resting state.
Rhodopsin transforms the absorption of light into a physiological signal through conformational changes that activate the intracellular G protein transducin—a member of the Gi/o/t family—initiating a signaling cascade, resulting in electrical impulses sent to the brain and ultimately leading to visual perception. Although previous studies have provided valuable insights into the mechanism of signal transduction in rhodopsin, methods that provide both a high spatial and temporal resolution are necessary to fully understand the activation mechanism at the atomic scale from femtoseconds to milliseconds. This study presents the first experimentally-derived picture of the rhodopsin activation mechanism at the atomic scale using time-resolved serial femtosecond crystallography in association with hybrid quantum mechanics/molecular mechanics (QM/MM) simulations. The results show that light-induced structural changes in rhodopsin occur on a timescale of hundreds of femtoseconds, and they reveal new details about the conformational changes that occur during activation.
Quantum disordered ground state in the triangular-lattice magnet NaRuO2
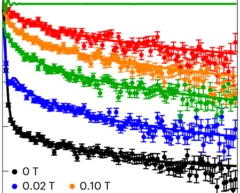
It has long been hoped that spin liquid states might be observed in materials that realize the triangular-lattice Hubbard model. However, weak spin–orbit coupling and other small perturbations often induce conventional spin freezing or magnetic ordering. Sufficiently strong spin–orbit coupling, however, can renormalize the electronic wavefunction and induce anisotropic exchange interactions that promote magnetic frustration.
When electrons dress up in light
Light – the Fifth Element of Materials Science?
Active learning-assisted neutron spectroscopy with log-Gaussian processes
Neutron scattering experiments at three-axes spectrometers (TAS) investigate magnetic and lattice excitations by measuring intensity distributions to understand the origins of materials properties. The high demand and limited availability of beam time for TAS experiments however raise the natural question whether we can improve their efficiency and make better use of the experimenter’s time.
Quality control of future transistors: Tackling the challenge of looking at atoms buried in silicon without moving them
Tackling the challenge of looking at atoms buried in silicon without moving them
X-rays make 3D metal printing more predictable
Insights into the microscopic details of 3D printing from the Swiss Light Source SLS could propel the technology toward wider application
SDU: Software for high throughput automated data collection at SLS MX
The Smart Digital User (SDU) software for unattended data collection has been deployed at the macromolecular crystallography beamlines at the Swiss Light Source.
Calcium sensor helps us to see the stars
New insight into how the protein calmodulin interacts with an ion channel in the eye could explain how our eyes achieve remarkable sensitivity to dim light.
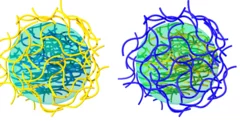
Amyloid-polysaccharide interfacial coacervates as therapeutic materials
Coacervation via liquid-liquid phase separation provides an excellent oppor- tunity to address the challenges of designing nanostructured biomaterials with multiple functionalities. Protein-polysaccharide coacervates, in particular, offer an appealing strategy to target biomaterial scaffolds, but these systems suffer from the low mechanical and chemical stabilities of protein-based condensates. Here we overcome these limitations by transforming native proteins into amyloid fibrils and demonstrate ...
Mobility of Dissolved Gases in Smectite under Saturated Conditions
Mobility of water, sodium and gas molecules within a smectite nanopore
Various gases are produced by metal corrosion and organic material degradation in deep gelological repository for nuclear waste. To ensure repository safety, it's important to demonstrate that gases can be dissipated by diffusion in host rocks and prevent pressure buildup in repository near field. Smectite mineral particles form a pore network that is usually saturated with water, making gas diffusion the primary transport mechanism. Molecular simulations have shown that the diffusion of gases through the pore network depends on various factors, including pore size and temperature. For instance, smaller pores and lower temperatures tend to reduce gas diffusion. Interestingly, hydrogen and helium have been found to diffuse faster than argon, carbon dioxide, and methane, possibly due to interactions with the clay surface and water molecules. Ultimately, the diffusion coefficients for different gases and pore sizes can be predicted using an empirical relationship, which is useful for macroscopic simulations of gas transport.