Scientific Highlights
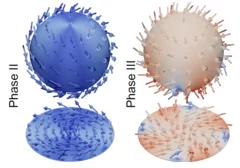
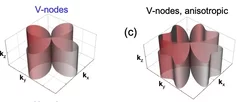
Skyrmion metamorphosis: Lattice transitions of hybrid skyrmions in a polar magnet
Magnetic skyrmions, with their distinctive vortex-like magnetic spin configurations, continue to intrigue researchers due to their potential applications in nanoscience and technology. Traditionally skyrmions form two-dimensional hexagonal close-packed lattices, with the skyrmions themselves displaying one of just two types of internal magnetization texture known as Bloch- or Néel-type. Recent theories hinted at the prospect of reconfigurable transitions between skyrmion phases of different lattice types and internal textures. Until now, experimental evidence supporting such theories has been scarce.
In-Plane Magnetic Penetration Depth in Sr2 RuO4 : Muon-Spin Rotation and Relaxation Study
We report on measurements of the in-plane magnetic penetration depth (λab) in single crystals of Sr2RuO4 down to ≃0.015 K by means of muon-spin rotation-relaxation. The linear temperature dependence of λ−2ab for T≲0.7 K suggests the presence of nodes in the superconducting gap. This statement is further substantiated by observation of the Volovik effect, i.e., the reduction of λ−2ab as a function of the applied magnetic field. The experimental zero-field ...
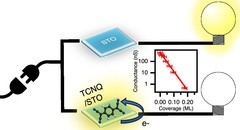
Switching Off the Surface Conductivity of Strontium Titanate by Non-Volatile Organic Adsorbates
Strontium titanate is a wide band gap semiconductor. Its surface can be rendered conductive by a mild annealing in vacuum. The present study reveals that by deposition of less than a monolayer of non-volatile organic molecules such as tetracyanoquinodimethane (TCNQ) this conductivity can be completely turned off. In view of the small size of TCNQ (ca. 1 nm) this could allow new pathways toward oxide-based electronics.
The secret life of an electromagnon
SwissFEL sheds light on how lattice and atomic spins jiggle together.
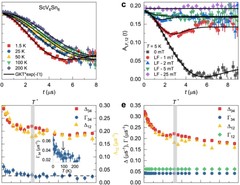
Hidden magnetism uncovered in a charge ordered bilayer kagome material ScV6Sn6
Charge ordered kagome lattices have been demonstrated to be intriguing platforms for studying the intertwining of topology, correlation, and magnetism. The recently discovered charge ordered kagome material ScV6Sn6 does not feature a magnetic groundstate or excitations, thus it is often regarded as a conventional paramagnet. Here, using advanced muon-spin rotation spectroscopy, we uncover an unexpected hidden magnetism of the charge order. We observe an enhancement ...
Microstructural control of additively manufactured Ti-6Al-4V
In-situ Selective Laser Heat treatment is applied to induce martensite decomposition in Ti-6Al-4V
Terbium-161: new radionuclide therapy hits the clinic
Highly targeted cancer treatment has the potential to eliminate ultra-small cancer lesions that cause disease recurrence.
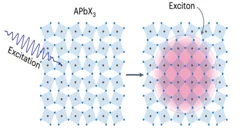
Excitons coupling to octahedral tilts in Pb nano-perovskites
Excitons coupling to octahedral tilts in Pb nano-perovskites
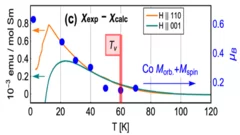
Crystal field rules heavy fermion delocalization in SmCoIn5
Crystal field rules heavy fermion delocalization
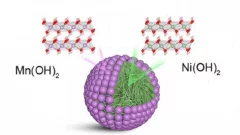
"Core-shell" cathodes for high performance Li-ion batteries
“Li-rich Ni-rich” core-shell particles are engineered as layered cathode materials for high energy Li-Ion batteries, including a controllable outer "Li-rich Mn-rich" shell improving cyclability.