Energy and Environment Research Division
Research at PSI comprises all aspects of human energy use, with the ultimate goal of promoting development towards a sustainable energy supply system. Technologies are being advanced for the utilization of renewable energy sources, low-loss energy storage, efficient conversion, and low emission energy use. Experimental and model-based assessment of these emissions forms the basis of a comprehensive assessment of economic, environmental and social consequences, for both present and future energy supply systems.
Division Head: Prof. Dr. Thomas Justus Schmidt
Energy Briefing Event 2023
The Energy and Environment Division of the Paul Scherrer Institut PSI successfully hosted their second Energy Briefing Event at the Zentrum Paul Klee in Bern. The event focused on the potentials and challenges associated with the production, regulation, and utilization of synthetic fuels. Representatives from WWF, Avenergy, PSI, and BAZL shared their expertise and insights on this topic.
A heartfelt appreciation goes out to Ulrich Koss (Metafuels), Theo Rindlisbacher (BAZL), Christian Bach (Empa), Thomas J. Schmidt (PSI), Thomas Häusler (WWF Switzerland), Daniel Hofer (Avenergy Suisse), and our moderator Stephan Lendi for their invaluable contributions and insightful perspectives.
Energy Briefing Event 2022
On June 28th, 2022, the Energy Divisions (ENE and NES) at PSI hosted their first Energy Briefing Event at the Kursaal in Bern. Knowledgeable voices from industry, research and government shared insights in a dialogue on the feasibility of the Net Zero goal and what next steps are required to achieve this collectively.
A big thank you to Daniela Decurtins (GazEnergy), Particia Sandmeier (Hitachi Energy), Martin Naef (ABB), Pascal Previdoli (BFE), Thomas Schmidt (PSI), Christian Verhoeven (GE), Peter Richner (Empa), Andreas Pautz (PSI) and our Moderator Stephan Lendi for their valuable contributions and insights!
Highlights & News
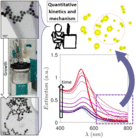
Kinetics and Mechanism of Metal Nanoparticle Growth via Optical Extinction Spectroscopy and Computational Modeling: The Curious Case of Colloidal Gold
An overarching computational framework unifying several optical theories to describe the temporal volution of gold nanoparticles (GNPs) during a seeded growth process is presented. To achieve this, we sed the inexpensive and widely available optical extinction spectroscopy, to obtain quantitative kinetic data. In situ spectra collected over a wide set of experimental conditions were regressed using the hysical model, calculating light extinction by ensembles of GNPs during the growth process. This model rovides temporal information on the size, shape, and concentration of the particles, and any electromagnetic interactions between them. Consequently, we were able to describe the mechanism of GNP growth and divide the process into distinct genesis periods. We provide explanations for several longstanding mysteries, e.g., the phenomena responsible for the purple-greyish hue during the early stages of GNP growth, the complex interactions between nucleation, growth and aggregation events, and a clear distinction between agglomeration and electromagnetic interactions. The presented theoretical formalism has been developed in a generic fashion so that it can readily be adapted to other nanoparticulate formation scenarios such as the genesis of various metal nanoparticles.
Chemically mapping ice forming particles
Scientists have just nucleated ice in an X-ray microscope for the first time and they created chemical maps of those responsible.
2018 Highly Cited Researchers
Three LAC researchers were highly cited in 2018.
Breakthrough - electron crystallography for everyone
Recent advances in electron crystallography published in Angewandte Chemie and highlighted by Science, Chemical & Engineering News and ScienceNews!Under the lead of LSK member, "Rapid structure determination of microcrystalline molecular compounds using electron diffraction", published in Angewandte Chemie International Edition https://onlinelibrary.wiley.com/doi/abs/10.1002/anie.201811318 has attracted great attention in the chemistry community.
Why the Little Ice Age ended in the middle of the 19th century
In the first half of the 19th century, a series of large volcanic eruptions in the tropics led to a temporary global cooling of Earth's climate. That Alpine glaciers grew and subsequently receded again during the final phase of the so-called Little Ice Age was due to a natural process. This has now been proven by PSI researchers on the basis of ice cores.
Brennstoffzellen zum Durchbruch verhelfen
Wasserstoff gilt als vielversprechende Alternative für eine Zukunft ohne fossile Energieträger. Um Brennstoffzellen weiterzuentwickeln und für einen Markteintritt vorzubereiten, verstärkt die Empa die Zusammenarbeit mit der H2 Energy Holding AG und dem Paul Scherrer Institut (PSI).
Light from the particle accelerator helps to understand ozone decomposition
PSI researchers have developed an experimental chamber in which they can recreate atmospheric processes and probe them with unprecedented precision, using X-ray light from the Swiss Light Source SLS. In the initial experiments, they have studied the production of bromine, which plays an essential role in the decomposition of ozone in the lower layers of the atmosphere. In the future, the new experiment chamber will also be available for use by researchers from other scientific fields.
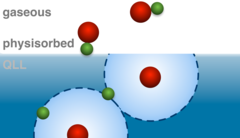
Coexistence of Physisorbed and Solvated HCl at Warm Ice Surfaces
The interfacial ionization of strong acids is an essential factor of multiphase and heterogeneous chemistry in environmental science, cryospheric science, catalysis research and material science. Using Near Ambient Pressure X-ray Photoelectron (NAPP) spectroscopy, we directly detected a low surface coverage of adsorbed HCl at 253 K in both molecular and dissociated states and interpret the results as physisorbed molecular HCl at the outermost ice surface and dissociation occurring upon solvation deeper in the interfacial region. This study gives clear evidence for nonuniformity across the air−ice interface and questions the use of acid−base concepts in interfacial processes.
Gasoline cars produce more carbonaceous particulate matter than modern filter-equipped diesel cars
In contrast to nitrogen oxides, modern gasoline cars emit much more cancerogenic primary soot (black carbon + primary organic aerosol) and lead to more toxic secondary organic aerosol than modern diesel vehicles.