Energy and Environment Research Division
Research at PSI comprises all aspects of human energy use, with the ultimate goal of promoting development towards a sustainable energy supply system. Technologies are being advanced for the utilization of renewable energy sources, low-loss energy storage, efficient conversion, and low emission energy use. Experimental and model-based assessment of these emissions forms the basis of a comprehensive assessment of economic, environmental and social consequences, for both present and future energy supply systems.
Division Head: Prof. Dr. Thomas Justus Schmidt
Energy Briefing Event 2023
The Energy and Environment Division of the Paul Scherrer Institut PSI successfully hosted their second Energy Briefing Event at the Zentrum Paul Klee in Bern. The event focused on the potentials and challenges associated with the production, regulation, and utilization of synthetic fuels. Representatives from WWF, Avenergy, PSI, and BAZL shared their expertise and insights on this topic.
A heartfelt appreciation goes out to Ulrich Koss (Metafuels), Theo Rindlisbacher (BAZL), Christian Bach (Empa), Thomas J. Schmidt (PSI), Thomas Häusler (WWF Switzerland), Daniel Hofer (Avenergy Suisse), and our moderator Stephan Lendi for their invaluable contributions and insightful perspectives.
Energy Briefing Event 2022
On June 28th, 2022, the Energy Divisions (ENE and NES) at PSI hosted their first Energy Briefing Event at the Kursaal in Bern. Knowledgeable voices from industry, research and government shared insights in a dialogue on the feasibility of the Net Zero goal and what next steps are required to achieve this collectively.
A big thank you to Daniela Decurtins (GazEnergy), Particia Sandmeier (Hitachi Energy), Martin Naef (ABB), Pascal Previdoli (BFE), Thomas Schmidt (PSI), Christian Verhoeven (GE), Peter Richner (Empa), Andreas Pautz (PSI) and our Moderator Stephan Lendi for their valuable contributions and insights!
Highlights & News
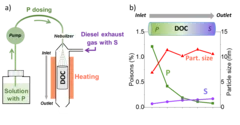
Unravelling the catalyst aging phenomena in vehicle emission control.
PSI has collaborated with catalyst and engine manufacturers to understand the aging phenomena of emission control catalysts. To this end, a diesel oxidation catalyst with a relevant mileage was carefully analysed; the results suggest that a complex combination of poisoning and thermal sintering is the cause of deactivation during driving. A reactor setup was then developed to simulate poisoning and sintering effects for prediction of catalyst durability in time and cost effective manner.
Two PSI-Scientists nominated «highly cited researchers»
In the research field “Geosciences”, 143 highly cited researchers were appointed worldwide this year, 7 of them from Switzerland, 2 of them from the Paul Scherrer Institute.
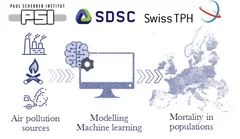
AURORA: from Air pollUtion souRces tO moRtAlity
The Laboratory of Atmospheric Chemistry has initiated innovative data-science-based modelling approaches to discover the most important pollution sources for human health
Blue hydrogen can help protect the climate
The key is to eliminate methane leaks.
PSI Impuls Award Winner Tobias Schuler
On November 25th, Tobias Schuler was awarded the PSI Impuls Award for his doctoral thesis titled ‘Towards a Generic Understanding of Porous Transport Layers in Polymer Electrolyte Water Electrolysis’.
PSI maintains its leading role in the CLOUD experiment at CERN
The CLOUD experiment at CERN will be recreating particle formation in key regions of the globe to understand the effects of these particles on regional climates
Discovery of new chemistry on aerosol surfaces
In a study published in Science, ambient-pressure X-ray photoelectron spectroscopy reveals spontaneous redox chemistry at solvating particle surfaces.
Carbon dioxide can be turned into a valuable resource
New study explores methods for using waste gas efficiently.
Take a deep breath. But how clean was the air?
The EU project, RI-URBANS, aims to improve air quality monitoring across Europe