Facilities in the Division of Biology and Chemistry
Electron Microscopy Facility at PSI (EMF)
The electron microscopy facility at PSI is presently sustained by three divisions, BIO (chair), NES and ENE. With its three transmission electron microscopes, a focused ion beam instrument and plenty of preparation tools it offers a direct access to electron microscopic techniques highly required in their fields of research (biological as well as materials science samples). The facility is unique within Switzerland as it also allows the investigation of radioactive materials.
Our state-of-art cryo-EM allows for structural analysis of biological macromolecules and organelles. Cryo-EM provides images of biological macromolecules and organelles in intact hydrated states. Computational image analysis enables three-dimensional reconstruction.
For more information please follow this link or contact Dr. Elisabeth Müller.
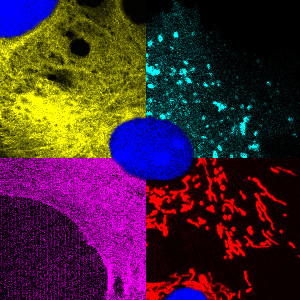
Light Microscopy
The Lab of Nanoscale Biology (LNB) is running the optical microscopy facility of the PSI Bio Division.
For more information please follow this link or contact Dr. Philipp Berger.
SwissFEL Biolab
We support serial crystallography experiments at SLS and SwissFEL with sample and injector expertise. Our aim is to help users to make the best out of the available beamtime as we believe a careful preparation is key for a successful experiment.
For more information please follow this link.
Protein Crystallography
A distinctive advantage of LBR as a research environment is the presence, on campus, of the Swiss Light Source (SLS). The macromolecular crystallography (MX) beamlines perfectly complement the excellent infrastructure of LBR for protein preparation, crystallization, crystallographic computing and structure analysis. We work in close collaboration with the MX group at the SLS in several areas, especially crystallization, in situ diffraction screening and data acquisition.
For more information follow this link or contact Dr. May Marsh.
Electrophysiological methods and devices
Translocation of ions and charged molecules across biological membranes are sensitively measured using electrophysiological instruments.
For more information please follow this link or contact Dr. Jinghui Luo.
Facilities at the Center for Radiopharmaceutical Sciences (CRS)
GMP Facility
In the CRS there is a state-of-art GMP facility for the production of radiopharmaceuticals under good manufacturing practice (GMP) for clinical applications.
For more information, please follow this link or contact Susanne Geistlich.
PET/SPECT/CT
The CRS has a preclinical nuclear imaging facility in house. Both, single photon emission computed tomography (SPECT), and positron emission tomography (PET) with an anatomic imaging modality by computed tomography (CT). These instrumentations give us the ability to analyze and visualize biologically significant events (such as tumor growth, hypoxia, angiogenesis, apoptosis) in combination with the anatomical information provided by CT.
For more information, please contact Dr. Martin Béhé.
LC/ESI-TOF-MS
The sensitivity and resolution of liquid chromatography/electrospray ionization time-of-flight mass spectrometry (LCT Premier Mass Spectrometer, Waters AG) allows the determining of mass of peptides or proteins.
For more information, please contact Alain Blanc.